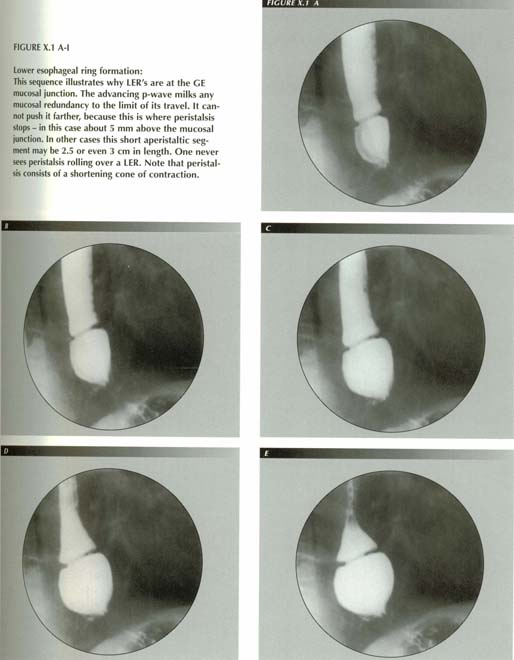
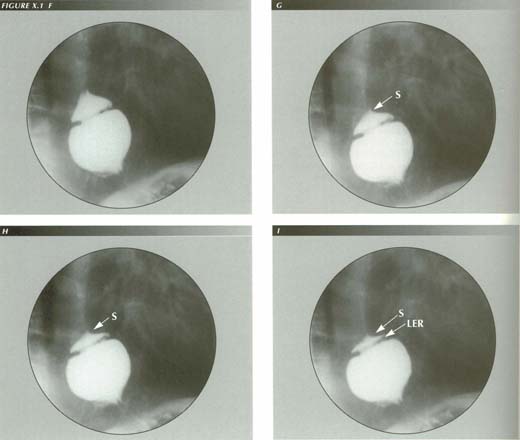
The lower esophageal ring
Although it was illustrated as a normal landmark by Templeton(1)
and first described in detail by Ingelfinger and Kraemer,(2)
the lower esophageal ring has become "Schatzki's ring" colloquially because
of a series of studies in which Schatzki and collaborators Gary(3)(4)
and MacMahon(5) described it in further detail,
reported the pathologic appearance at autopsy and correlated the symptoms with
the size of the ring.
These papers established that the ring occurs at the junction of stratified
squamous esophageal epithelium and gastric mucosa. It is invariably associated
with HH -- usually a sliding HH. It becomes symptomatic when the lumen is reduced
to about 1.9 cm. It varies in position from 4 to 5.5 cm above the diaphragm.
It is 2-4 mm in thickness. The lumen was reduced in one of their cases to as
little as 3 mm.
Schatzki and associates also noted that "If the esophagus distal to the narrowing
balloons out, as for instance during the Valsalva test, the ring seems to climb
away from the diaphragm and, conversely, it seems to migrate toward the diaphragm
when the distal esophagus collapses." (6)
This description would seem to show that they once believed that the fundic
pouch was "esophagus distal to the narrowing" yet in the same paper, they reported
that in 4 cases a change in the character of the mucosal pattern suggested "
. . . that the ring was at the herniated esophagogastric junction." Of course,
before the pathologic appearance had been described, it would be hard to be
certain that the ring was at the mucosal transition.
Brombart(7) soon ventured a respectful correction,
pointing out (incorrectly) that the deep inspiration of the Valsalva test accounts
for the withdrawing of the ring from the diaphragm and (correctly) that it is
not correct to speak of the segment below the ring as "esophagus" as it is the
intrathoracic portion of the stomach. Like Brombart and Schatzki, Templeton(8)
stated that the ring " . . . is brought out by distending the esophagus." -
also focusing on the distention while overlooking the maximal shortening that
then occurs at the end of the Valsalva maneuver.
Goldstein(9) has listed the various ideas concerning
the origin of the ring culled from his review of the literature as:
1.) Hypertrophied or dislocated constrictor cardia
2.) Overactive inferior esophageal sphincter
3.) Inflammatory stricture
4.) Cardiospasm
5.) Congenital malformation
6.) Trauma.
7.) In his 1970 review, Earlam(10) added mucosal
damage from reflux.
Various sphincters have also been proposed to explain the appearance. These
include:
8.) The "bracket bundles" of Ziano et al.,(11)
9.) The sphincter of Lerche(12) and
10.) The lower esophageal sphincter.
At present, he most popular rational for LERs is an undefined "inflammation."
It was inevitable that simultaneous manometric and cineradiographic examinations
would be performed to relate the ring to the sphincter. Surprisingly, 2 of the
4 placed the sphincter below the LER!(13),(14),(15),(16)
Such high-tech efforts have a flavor of overkill as simply looking at the radiographs
should convince one that the sphincter is above the ring.
MacMahon, Schatzki and Gary were the first to report that the mucosal transition occurred on the lower surface of the ring. The histologic findings they described, however, are not those of inflammation or fibrosis, ruling out Nrs. 3 and 7; the ring proved to be 2 layers of esophageal stratified squamous epithelium. Even the submucosa was not a part of the ring. It split into 3 layers without extending to the free edge of the ring. Subsequently, numerous biopsies, at both thoracotomy and endoscopy, have confirmed the initial pathologic description in several significant details. For example, in 14 biopsies, all obtained at thoracotomy, Postlewait and Musser(17) never found any annular muscle in edge of the web, thus disposing of the idea that it was a contractile ring (Nrs. 4, 8, 9 and 10). It is well established that:
The upper surface is squamous epithelium,
The lower surface is gastric epithelium, sometimes with "dribbles" of squamous
epithelium running over the edge.(18)
The muscularis mucosae is split into 3 layers,
The core of the web is usually filled with fibrous connective tissue although
the amount varies.
There are several additional features of these rings that are significant
and must be explained by any proposed theory of their pathogenesis:
Even a marked ring may be invisible at esophagoscopy
On followup, the ring may have diminished in size.
A single ring may break up into multiple rings or vice versa.
The ring may grow from mere notches to a definite ring as the esophagogastric
junction distends.
The ring may vanish when the sphincter fails at the end of a P-wave and regurgitation
from the HH to the distal esophagus occurs.
They are seldom seen with the huge herniations that occur with rupture of
the PEL.
They usually, but not invariably, disappear after a HH repair.(19)
In 1963, impressed by the latter fact, I suggested(20)
that the ring was redundant mucosa milked to the lower end of the esophagus
by the peristaltic wave. Schatzki did not agree.(21)
According to Ott et al.(22) this mechanism(23)
is now the main competing theory with inflammation. As was demonstrated in Chapter
2, however, mucosal redundancy is a consequence of LM contraction. The esophagus
is only an exception to the general rule because its length is fixed by its
upper and lower attachments. If the PEL is stretched - that is, when there is
a hiatal transtract - the elasticity of the mucosa and contraction of the m.
mucosae cannot take up the slack and a fold must form. The invariable
association of gastric transtraction and LERs is because, in order to create
enough mucosal redundancy to form a ring, the esophagus must shorten enough
to pull the fundus through the hiatus.
Like a mucosal fold, a LER can come and go and accordion-like pleats can form in various configurations. One can sometimes see this happen in rapid sequence or cine frames. This is not the case with congenital malformations or strictures. The redundancy may form multiple fine folds or be gathered up into a large, deep fold. One patient had a single deep ring, yet presented with 3 shallow rings a year later.
Mucosal folds do, however, tend to display a marked constancy. The same physical
circumstances that caused a fold to form in one way the first time operate,
ceteris paribus, to repeat that configuration the next time. Moreover,
living tissues tend to take a set, as though they were graven in stone ultimately,
due to the microscopic fine structure that changes but slowly over time.
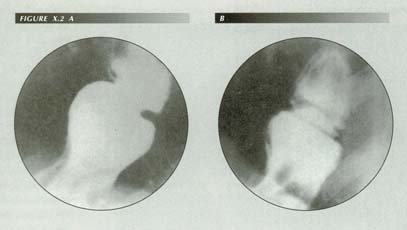
(A) A typical deep ring can break up into several shallow rings. (B) Roentgenogram made nearly a year after A. Note three fine �accordion pleats� at the cardioesophageal junction. Reprinted from Stiennon, O. Arthur AJR:90 811-22, 1963 with permission.
The growth of the ring with distention of the phrenic ampulla is due, not to
distention per se, but to the shortening of the esophagus that occurs
concurrently with that distention. If the ring consisted of an annular band
of fibrosis, for example, distention would then produce a ring where none existed
in the collapsed organ. However, in that case the band would be in the free
edge of the ring on histological section. But this is not the case. Although
there is fibrotic tissue in the core, the amount varies from slight to striking
and is distributed in a wedge, not in a core at the free edge.
It is not intuitively obvious, but consider that a fold has formed that is
prominent enough to narrow the lumen to 1 cm. If the esophagus in the region
of the fold then dilates another millimeter, it will take 2 mm of slack to keep
the ring just as narrow as it was before. Allowing for, say, 15% elasticity
the esophagus must shorten more than 2 mm to produce the required mucosal redundancy.
This is the reason that as the ring forms, it does not seem to get deeper.
Of course, the "climb away from the diaphragm" during a Valsalva test noted
but not explained by Schatzki and Gary is due to LMC with esophageal shortening.
These 2 factors - a relaxed PEL and a shortened esophagus - are the factors
that produce HH. The association of rings with HH occurs because they are both
caused by the same thing - LMC. After a repair, especially a pulldown procedure,
this ability to shorten unduly is overcome and the redundancy cannot form. This
results in disappearance of the ring.
In the intervening 32 years, the suggested mechanism has held up well when
tested against day to day experience with one still puzzling feature: further
experience showed that not all rings were cured by a HH repair. One has to assume
that, in such cases at least, the two surfaces of the ring became adherent so
that elongating the esophagus will no longer obliterate the fold. In an occasional
case,(24) submucosal fibrosis is a striking
finding. It would seem that such rings must be of the type that can survive
an effective HH repair.
Multiple LERs: Can each one have a different etiology? Or are they accordion pleats?
LERs are a splendid example of the general rule formulated in Chapter 2: folds
are orthogonal to the muscular fibers that cause them. Thus, the rule is
as useful in finding the cause of a fold as it is in predicting how it will
be arranged. Here, a transverse fold implies LMC as, for that matter, do reflux
and HH - the usual concomitants of LER's.
Perhaps because it first called my own attention to the LM, the circumstance that most LER's vanish after a gastropexy of the Boerema type, seems to me the most conclusive evidence for the plication mechanism. How else can one explain the clearing of a pathologic finding when there has been no local attack on it? Elongating the esophagus by a pulldown procedure restores esophageal length, removes the mucosal redundancy and the pleat can no longer form.
The appearance and disappearance of the rings during a radiological examination
has been explained by postulating that distention of the ampullary region is
necessary to demonstrate them.(25) But this
distention is normally achieved by the Valsalva maneuver. Obstruction in the
PEL tent forces distention when the bolus cannot pass the sphincter. The region
can be distended in other ways e.g., by performing a double contrast esophagogram
or by rapid swallowing. If these do not produce shortening, a ring will not
appear.
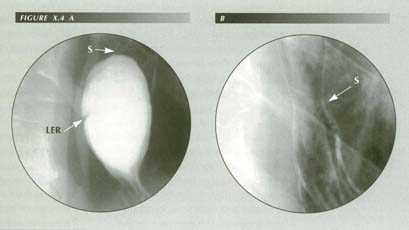
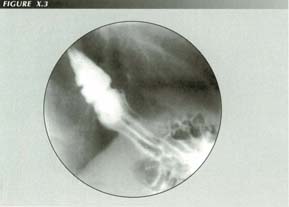
Disappearing LER: When the HH is demonstrated with the Valsalva maneuver (A), a typical LER forms. When it is evoked by inducing belching (B) there is no trace of a ring. The main difference is that there was no peristaltic wave in (B) to milk mucosal redundancy distally. Also note the long aperistaltic segment between ring and sphincter. The slight hourglass constriction in (B) (arrow) is the less distensable sphincter region. This may be misinterpreted as a stricture in an air distended esophagus and be diagnosed Barrett�s esophagus.
Both manometry and radiography by custom only display the deglutition of fluids,
for which peristalsis is virtually redundant. Eating, however, is concerned
with the ingestion of semi-solid, particulate masses of varying size and consistency.
A 2.5 cm marshmallow, for example, can be swallowed whole. Imperfectly masticated
particles of meat of this size are sometimes ingested accidentally. For such
to clear the esophagus, peristalsis is imperative.
For this reason, patients learn to eat slowly and chew their food well to avoid
obstructive episodes, not by large food particles themselves but by the
maximal LMC they provoke. If esophageal transit is mainly by gravity flow,
the LM contracts only slightly. If swallowing is against resistance, LMC and
esophageal shortening is maximal provoking the ring.
The fact some rings of great chronicity persist after a HH repair suggests
that local inflammation and fibrosis eventually fuse the two surfaces of the
ring. Perforating veins will be ruptured when the ring first forms. The m.
mucosae is split, as MacMahon et al. noted, a portion of it following
the superior surface of the ring, a portion extending into the core of the ring
and a portion continuing to form the m. mucosae of the stomach. Slight
hemorrhage easily explains the usual residual of a few lymphocytes and fibrosis.
I am not aware that proponents of other etiologies have explained this splitting
of the m. mucosae. In the case of fold formation, the reason for the
split is obvious: once the lamina propria folds, it no longer follows the muscular
wall of the organ and must sever its connection at some point. The cleavage
is in the m. mucosae with some fibers adhering to the wall, some to
the lamina propria and others simply ruptured.
The location of the ring at precisely the squamo-columnar junction can scarcely
be fortuitous. The milking function of the peristaltic wave ends at the sphincter
so the mucosal redundancy cannot be milked down into the stomach and lost. On
the other hand, the p-wave doess not stop short of the sphincter. This is why
these rings are not located proximally. The location of the ring below the sphincter
is further proof of the pathogenesis and simultaneously a verification of the
extent of the p-wave.
Because many rings are not fixed, this milking action of peristalsis may be essential to their formation. When the conventional full column study with Valsalva test is performed the detection rate according to Ott's group(26) was 97% whereas the double contrast method only detected 58% of the 60 rings they studied. The obvious difference is that there is no milking p-wave when air contrast is used. The 39% difference suggests an order of magnitude for the percentage of unfixed rings, however, the same group, in an earlier study,(27) could find only 17% of proven rings with air contrast, suggesting that most rings are not fixed.
The lower detection rate with endoscopy in the Bowman Grey series (35 of 58)
reinforces this argument. The scope also elongates the esophagus rather than
shortening it and, of course, there is no peristalsis during the examination.
Does inflammation cause the LER?
Because there is frequently reflux and esophagitis associated with HH and
consequently with rings, inflammation has been an attractive hypothesis as an
explanation of ring formation. However, this is post hoc ergo propter hoc
reasoning. If one tries to see how inflammation could cause the
thin, web-like narrowing, he looks in vain for support from the histologic findings.
Typically there are a few lymphocytes and plasma cells but rarely a polymorphonuclear
cell.
Surgically, the webs cannot be palpated without opening the stomach. One would
expect obvious induration if inflammation were present. Endoscopically, two
thirds of the cases will show evidence of mild to moderate esophagitis. This
does not explain the genesis of LER's in the third that have no esophagitis.
Many patients with LERs have no reflux symptoms.(28)
Nowhere else in the GI tract does inflammation cause webs or rings. On the
contrary, it causes long hourglass constrictions. Moreover, the appearance of
inflammatory disease in the lower esophagus has already been spoken for by esophagitis.
Superficial inflammation causes enlarged longitudinal folds to form.
The same mechanism cannot be used to explain both longitudinal and transverse
folds.
When inflammation is deep, the appearance is that of an inflammatory stricture.
Inflammatory strictures invariably have a longitudinal extent several times
the diameter of the organ they involve, whether it be the esophagus, Fallopian
tube, urethra, ureter, large or small bowel. This is the exact opposite of the
situation seen with LER's. If one advances an inflammatory theory of ring formation
one must explain why this rule does not apply; why the ring can divide into
two or more rings; why the submucosa is not involved; why a pulldown procedure
obliterates them; why they occur where they occur; how the same cause can have
two effects and so on.
Of course, inflammation cannot explain the choice of the mucosal junction as the location of the rings. When a HH is repaired by pull-down, the ring - if it disappears at all - vanishes at the operating table. This is not the slow resolution typical of inflammatory disease. A recurrence of the ring is associated with a recurrence of the HH(29) and reflux without recurrence does not reproduce it.
The demise of the two-pouch theory of the esophagus
One consequence of the LER and its definition as the true esophago-gastric
mucosal junction is the downfall of the two-pouch theory. According to this
there are a variety of dilatations of the lower esophagus. On one of these there
is widespread agreement. This is the dilatation proximal to the ring that corresponds
with what Templeton(30) called the "phrenic
ampulla." Distal to the ring, variously identified as the gastroesophageal vestibule
(by Lerche), the Vormagen of Arnold, the cardiac antrum of Luschka, the epiphrenic
bell and the abdominal gullet favored by several British authors.
The mere fact that all these dilatations are below the ring means that they
are lined with gastric mucosa. Without further evaluation this allows us to
dismiss not only these "structures", but the large body of opinion, doctrine
and speculation on which much of the treatment of disorders of the area is based.
They are all unrecognized sliding transtractions.
That competent investigators accepted these interpretations for so long points
up an interesting feature of HH: LMC pulling the fundus through the hiatus can
convert it from a hemispherical to a tubular shape. This tube looks enough like
the esophagus to mislead several generations of radiologists and anatomists.
The distorted part of the fundus can take a set so that its tubular shape persists
even after the HH that caused it has reduced. In this state, it was seen post
mortem by the anatomists as it can occasionally be seen radiologically.
It is noteworthy that, although rings come and go with LM contraction, they
nearly always reform in the same place. The preformed split in the m. mucosae
facilitates the reformation in the same site rather than at a new one.
As a tissue is torn in the process and perforating veins ruptured, it is not surprising to find some evidence of round cell infiltration and not the profuse polymorph infiltrate characteristic of inflammation.
SUMMARY
LER's are transverse folds caused, like all folds, by contraction of the orthogonal
component of the muscularis propria - in this case the LM of the esophagus.
Its contraction causes mucosal redundancy that is milked as far distally as
the p-wave can carry it, that is just beyond the LES.
Although HH, reflux and esophagitis are all seen with LER's, they neither
cause the ring nor are they caused by it. The entire constellation of findings
is caused by LMC.
Elongating the esophagus will cure the ring by removing the redundancy unless the two surfaces become adherent. The details of the histologic structure of the rings are only consistent with a plication mechanism.
References
Last Updated July 27, 2007 by David PJ Stiennon
1. 1.Templeton, Fredrick E., X-ray Examination of the Stomach. University of Chicago Press, Chicago, 1966.
2. . Ingelfinger, F.J. and Kramer, P., Dysphagia produced by contractile ring in lower esophagus, Gastroenterology 23:419-30, 1953.
3. 3. Schatzki, R., and Gary, J.E. Dysphagia due to a diaphragm-like localized narrowing in lower esophagus ("lower esophageal ring"). Amer. J. Roentgenol. Rad. Therapy & Nuclear Med. 70:911-22, 1953.
4. 4. Schatzki, R. and Gary, J.E. Lower esophageal ring. Amer. J. Roentgen. Rad. Therapy & Nuclear Med. 84:1142-1143, 1956.
5. 5. MacMahon, H.E., Schatzki, R. and Gary, P. Pathology of the lower esophageal ring; report of case with autopsy, observed for nine years. New England J. Med. 259:1-8, 1958.
6. Inadvertently, as it were, they were describing LM contraction. A good example of my thesis that even first class observers have not seen LM activity.
7. 6. Brombart, Marcel, Idem.
8. 7. Templeton, Fredrick E., Lower esophageal ring. New Eng. J. Med. 283: ,1970.
9. 8. Goldstein, Franz, Dysphagia with iron deficiency, esophageal webs and esophageal rings. In Bockus.
10. 9. Earlam, Richard, Clinical tests of oesophageal function, Grune & Stratton, New York, 1975.
11. 10. Ziano, C., Poppel, M.H., Jacobson, H.G., Lepow, H and Osturk, C.H. Lower esophageal vestibular complex; anatomic-roentgen study. AM. J. Roentgenol. Rad. Therapy & Nuclear Med. 84:1045-55, 1960.
12. 11. Lerche, W. The esophagus and pharynx in action. A study of structure in relation to function. Charles C. Thomas, Springfield, IL, 1950.
13. 12. Eckardt, V.F., Adami, B., Hucker, H. and Leeder, H., The esophagogastric junction in patients with asymptomatic lower esophageal rings. Gastroenterology 79:426-30, 1980.
14. 13. Harris, L.D. and Kelly, J.E., Jr. and Kramer, P., Relation of the lower esophageal ring to the esophagogastric junction.
15. 14. Cauthorne, R.T., Vanhoute, J.J. and Donner, M..W., Study of patients with lower esophageal ring by simultaneous cineradiography and manometry. Gastroenterology 49: 632-40, 1965.
16. 15. Heitman, P., Wolf, B.S. and Sokol, E.M., et al., Simultaneous cineradiographic-manometric study of the distal esophagus: small hernias and rings. Gastroenterology 50: 737-53, 1966.
17. 16. Postelwait, R.W. and Musser, A Wendell, Pathology of the lower esophageal web. SG&O 120:571-75, 1965.
18. If a LER were flattened out by stretching the esophagus, these dribbles would be indistinguishable from the interdigitations of gastric and squamous epithelium seen in the Barrett esophagus.
19. 17. Vansant, John H., Surgical significance of the lower esophageal ring. Ann. Surg. 175:733-9, 1972.
20. 18. Stiennon, O. Arthur The anatomic basis for the
lower esophageal contraction ring; plication theory and its applications. Amer.
J. of Roentgenol. Rad. Therapy & Nuclear Med. 90:811-22,
1963.
21. 19. Roessler, Marvin, Personal communication.
22. 20. Ott, David J., Gelfand, David W., Wu, Wallace C. and Castell, Donald O., The esophagogastric region and its rings. AJR 142: 289-92, 1984.
23. Which they attribute to later observers.
24. 21. Wilkens, E.W. Jr. and Bartlett, M.K., Surgical treatment of the lower esophageal ring. New Eng. J. Med. 268:461-4, 19
25. 22. Ott, David J., Gelfand, David W., Wu, Wallace C. and Castell, Donald O., 1984. Op cit.
26. 23. Ott, David J., Chen, Yu Men, Wu, Wallace C., et al., Radiographic and endoscopic sensitivity in detecting lower esophageal rings. AJR 147: 261-65, 1988.
27. 24. Chen, Yu Men, Ott, David J., Gelfand, D.W., et al., Endoscopic sensitivity in detecting lower esophageal rings. J. Clin. Gastroenterol. 7: 121-5, 1985.
28. 25. Levin, Marc S., In: Textbook of Gastrointestinal Radiology, Gore, Richard M., Levin, Mark S. & Laufer, Igor, Eds., W.B. Saunders, Philadelphia, 1994.
29. 26. Eastridge, Charles E., Pate, James, Q.W. and Mann, James A., Lower esophageal ring: experiences in treatment of 88 patients. Ann. Thorac. Surg. 37:103-7, 1984.
30. 27. Templeton, Fredrick E., X-ray Examination of the Stomach. University of Chicago Press, Chicago, 1966.