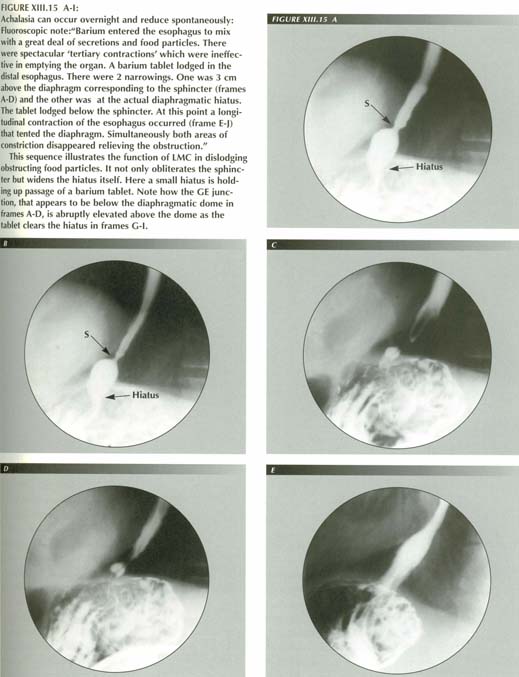
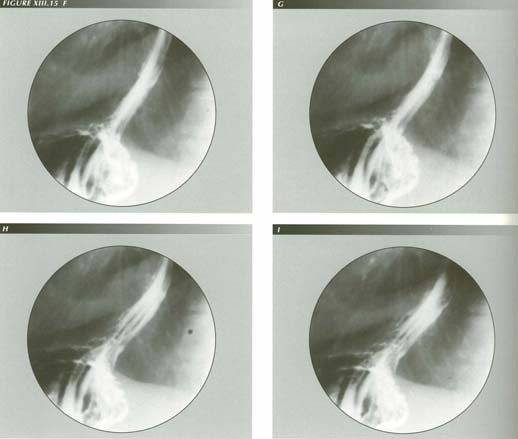
"Achalasia" and related misdiagnoses
It defies comprehension that a disorder causing such profound muscular hypertrophy
that it is treated like hypertrophic pyloric stenosis is still universally attributed
to a motor neuron deficit . This is equivalent to believing that the denizens
of Muscle Beach are paralytics.
On the face of this inconsistency it should be obvious that "achalasia," in
the conventional sense, is a misdiagnosis. The related "esophageal motor disorders"
(EMDs) that lead up to achalasia (including "idiopathic muscular hypertrophy")
must also be misdiagnoses. Our task is to arrive at the correct diagnosis. With
an appreciation of the cause of hiatus hernia to build on, we can do so. To
preserve for the reader the pleasure of discovery, I will depart from a principle
of exposition and not reveal the correct diagnosis up front.
Post poliomyelitits: The gluteal musculature in this patient with right sided paralysis shows what happens to aganglionic muscle.
Historical review
Early observers of achalasia took it for granted there would be increased tone
in the lower esophagus because a standing column of barium in the lumen well
above the diaphragm seemed to indicate a measure of resistance to outflow. Once
transducers and manometry became available, this simple concept was no longer
tenable: measurements with non-perfused catheters did not show the expected
elevation of pressure.
But the appearances persisted and had to be explained. Beginning in the 1940s,
those who followed Hurst and Rake(1) preferred
to refer to the condition previously known as "cardiospasm" as "achalasia."(2)
According to the new concept embodied in the name, the condition was not a spasm
of the sphincter but simply a failure of the sphincter to relax. "Failure of
relaxation" papered over the radiographic appearance and the manometric findings.
What to a radiologist looked like spasm of the sphincter, to the manometrist,
did not. Although a barium swallow was arrested at the diaphragm, a bougie or
esophagoscope passed through with "gentle pressure."
The new term was still unsatisfactory. While stating the obvious, like the
term it replaced, it implied that there was some intrinsic malfunction of
the sphincter. Although it inspired many studies of the pharmacology and
neurology of the sphincter, the implication was never established. The attention
thus directed to the sphincter reinforced the idea that the disease was an aganglionic
destruction of normal reflex control.
The idea of an aganglionosis arose from the finding, again by Hurst and Rake,
of a 90% reduction in the number of ganglion cells per low power field in sections
taken from the body of the esophagus. A 90% loss of ganglion cells could be
expected to interfere with the normal function of the delicate and complex submucosal
and myenteric plexuses - whatever that function might be. Unfortunately, once
the problem is pushed down to the cellular level, it becomes a problem for the
neurophysiologist.(3)
The aganglionic theory got additional support from the finding of Kramer and
Ingelfinger that the achalasic esophagus was hypersensitive to Mecholyl. According
to Cannon's law,(4) this also implied a denervated
end organ.
The aganglionic hypothesis found ready acceptance because of two analogs or
"models" - aganglionic megacolon and Chagas' disease.(5)
The strong radiologic resemblance between achalasia and aganglionic megacolon
- a narrow segment of gut with a grossly dilated gut behind it - supported the
hypothesis. Chagas' disease, an aganglionosis due to destruction of ganglion
cells by a trypanosome, produced an esophageal appearance identical with achalasia.
Apparently clinching the proof was the finding of degeneration of cells in the
dorsal motor nuclei of the vagus and the nucleus ambiguus, the centers for neurological
control of the esophagus.(6)
Thus, by 1969 Misiewicz et al.(7) could accurately state, "It is generally agreed that achalasia of the cardia is caused by degeneration of the myenteric nervous plexus." In a more recent review (1983), Wong and Johnson(8) found that the focus of current research is still on the neurologic changes but concluded that the underlying cause of these changes was unknown. A 1986 surgical monograph(9) includes achalasia in the chapter headed "Neurogenic Disturbances." Castell(10)
(1986,1992) indorses this view. Investigating the complex neuropharmacology(11)
remains a problem of great interest, however, because of the widespread conviction
that achalasia is caused by an aganglionosis.
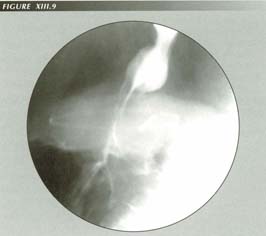
In the last twenty years, there have been nascent changes in the outlook on achalasia that, without refuting the aganglionosis theory, have begun to put the disease in a different light. Suspicion that diffuse esophageal spasm (DES) was intimately associated with achalasia was well documented in a significant single case report in 1967 by Kramer, Harris and Donaldson.(12) They followed a patient for eight years during which the clinical, radiological and manometric picture evolved from that of DES to achalasia. There is a still earlier report by Schroeder, et al.(13) recording the same transition and Barrett(14) also reported observing a transition from what appeared to be diffuse spasm to cardiospasm.
The significance of these cases was long in being recognized. In 1979 Vantrappen's
Belgian research group,(15) aware of the association,
attempted to define four classes of esophageal motility disorder (EMD) according
to the presence or absence of peristalsis and sphincter relaxation. These were
further subclassified as vigorous or non-vigorous, making eight groups in all.(16)
As a result of study of a large series, and perhaps as a way of sidestepping
the cardiospasm-achalasia-DES tangle of nomenclature, the all-inclusive term
"esophageal motor disorder" (EMD)(17) is now
employed for them all. This implies a belief they share a common, unknown, cause.(18)
Thus, there is reason to hope that, if we find a cause or cure for one, we have
found it for all.
It is probably fair to say, however, that the aganglionosis theory of achalasia
is still regarded as essentially correct(19)
or correct but incomplete. That it remains so entrenched after more than two
generations and an enormous number of clinical, surgical, radiological and physiological
studies, is a splendid exemplification of Kuhn's(20)
thesis that scientific research will always be interpreted in terms of the current
theory whether or not the theory is correct. Many camels have been swallowed
in the process.
Flaws in the aganglionic theory
On examination, the evidence for an aganglionic origin of achalasia proves
flawed. The lack of any difference in response to a broad range of sympathetic
drugs between muscle strips taken from normal and achalasic patients(21)
is unexpected and argues against an aganglionosis. Patients with achalasia do
not have autonomic nerve deficits elsewhere in the gut.(22)
Moreover, the analogy with aganglionic megacolon is patently false. "In Hirschprung's
disease there is usually a narrow segment with no ganglion cells, a transitional
zone with few cells, and proximal gut with normal neurons, dilated because of
the distal obstruction."(23),(24),(25)
This is precisely the reverse of the situation with achalasia in which the dilated
region is supposed to be aganglionic, and the sphincter area to have ganglion
cells.
Nor does the analogy with Chagas' disease withstand scrutiny. Padovan, Godoy
et al.(26) studied 24 patients with
Chagas' disease and found that the average resting LESP(27)
was three standard deviations below normal. Moreover, these patients
were less sensitive to pentagastrin than normal subjects. These results
are the exact opposites of the findings in achalasia. Such patients are reported
to have high resting LESP and are supersensitive to pentagastrin.(28)
Holloway, Dodds (29) most recently report resting
LESPs in achalasia five standard deviations above normal (41 mm Hg).
Both groups found average values of 20 mm Hg in their normal controls. The Holloway,
Dodds group found untreated achalasia patients to be supersensitive to pentagastrin
as did Orlando and Bozymski.(30) Animal studies
in Chagas' disease(31) suggest that aperistalsis
precedes incomplete LES relaxation.
Therefore, both "models," instead of supporting the aganglionic hypothesis,
clearly refute it. Moreover, on analysis the cell "reduction" turns out to be
a geometrical artifact. The reduction is expressed in cells per low power
field.(32) A microscopist is accustomed
to making judgements of relative frequency of cells by counting their number
per microscopic field. Although this method is valid as a rule, it grossly underestimates
the cell count when applied to an inflated and stretched organ.
As the esophagus dilates, ganglion cells get farther apart so that there are
fewer per microscopic field. Like dots on the surface of a balloon, as the balloon
inflates, there are fewer dots per square cm although the total number
of dots is unchanged. So accurate is the variance of the cell count with radius
that, given one, we can calculate the other with great accuracy. As this fact
is generally overlooked, the idea that achalasia is an aganglionic dysplasia
has survived for two generations to obfuscate any serious analysis before it
starts.
The geometry also explains why investigators who took sections from the undilated
sphincter region found no significant reduction in ganglion cells although,
in the opossum, for example, ganglion cells reach a nadir in the sphincter region.(33)
Under the conditions prevailing in esophageal dilatation, many muscle cells
are effectively denervated. Stretching neurons and their processes in two directions
to many times their normal length may exceed their elastic limit or even avulse
them from motor end organs. In addition, stasis and infection can more easily
reach the myenteric plexus to cause degeneration through the thinned esophageal
wall. These effects can account for the hyperreactivity to Mecholyl.
The hypersensitivity of the achalasic esophagus to cholinergic drugs is undeniable, but the conclusion drawn from this - that the cause of the disease is aganglionosis - does not necessarily follow. Many cases of DES also have a positive Mecholyl test, although DES is not an aganglionic disease. Pathologic studies(34),(35) have failed to show degeneration of the myenteric plexus in DES. The sensitivity seems to progress as DES tapers into achalasia. Making use of this fact, graded doses of Mecholyl have been used(36) to differentiate the two or to place a patient in the DES-achalasia spectrum.
While this demonstrates the kinship of the two conditions, it also shows that
the seeds of achalasia are already present before the presumed ganglionic degeneration
occurs. Indeed, with further progression of the disease, the hypersensitivity
disappears. This is attributed to degeneration of the muscle end organ with
late stage disease. It is equally valid to attribute degeneration of nerve cells
to the same cause.
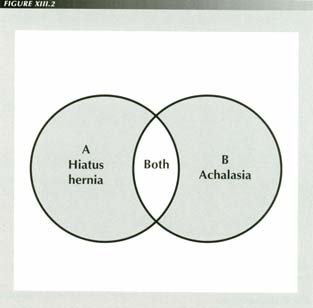
If the set of patients with both malady A and malady B is empty, i.e., ~(A&B), by the calculus of Boolean algebra this equals ~A or ~B. That is, patients are spared either one disorder or the other, but not both. This appears to be the case in practice: a patient can have achalasia or HH but not both. This implies that either one disease prevnts the other (which is obviously not true) or that they are the same disorder - now diagnosed one way, now the other.
Finally, the brainstem lesions are not primary. Commenting on the central
lesions, Earlam,(39) after reviewing the evidence
for trans-synaptic degeneration of postganglionic cells after destruction of
their afferent nerve supply, concluded that "there is absolutely no experimental
evidence that trans-synaptic degeneration actually occurs." Because it is unlikely
that well separated nuclei in the medulla would be affected bilaterally, Earlam
concluded it was " . . . far more likely that the central nuclei degenerated
after losing the neurons of the esophageal wall as retrograde degeneration is
well documented."(40) Dogs, the best studied
animal analogue, also have normal ganglion cells early in the disease.(41)
The ganglion cell degeneration observed is a fairly late effect, not
the cause of the disease.
Why are there no cases of HH and achalasia?
In this connection, the reported incompatibility of hiatus hernia and achalasia
becomes significant. Hiatus hernia is an extremely common radiologic finding.
It has been demonstrated radiographically in 100 consecutive cases! It would
be very surprising if many or most cases of achalasia were not accompanied by
hiatus hernia. Yet, in a review of the New Haven experience, Binder (42)
found that of 42 recorded cases of achalasia, only one was also reported as
having HH. On review of the exception, they concluded that diagnosis had been
erroneous.
HHs occur in association with every other esophageal disease -- with lower
esophageal rings (LER), tertiary contractions (TC), cardioesophageal reflux,
Zenker's diverticulum, scleroderma, carcinoma. It is an arresting circumstance
if they do not coexist with achalasia.
This exclusive or relationship has logical significance. If we ask,
"How would it be possible for an HH to preclude a diagnosis of achalasia in
the same individual?" The obvious answer is that they are the same thing! That
is, achalasia is simply a name for an HH with special characteristics
that prevent it from being recognized as such. This leads at once to the insight
that HHs are a factor in the pathogenesis of achalasia. We need only discover
the nature of those special characteristics. (The possibility that HH protects
one from achalasia can be dismissed.)
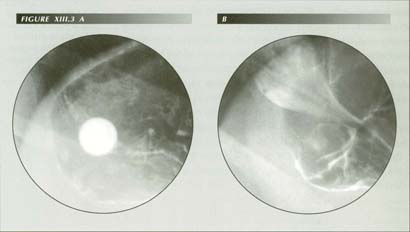
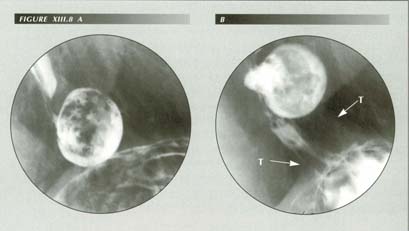
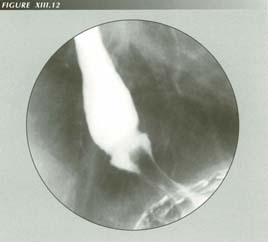
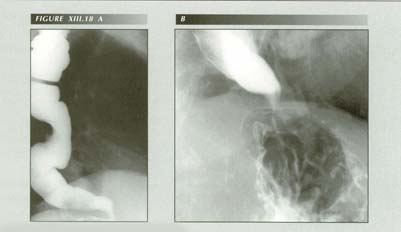
Hiatal canal obstruction: �Trouble swallowing food.� (A) A 12.5 mm barium tablet was held up at the hiatus. (B) this small hiatal canal was the cause of the obstrucion. Note the projection gives the false impression that the GE junction is below the diaphragm because of the slope of the latter.
More detailed examination of the morphology of the GE junction shows this
cannot be true. The distal esophagus can exist unobstructed in the hiatus because,
except for branches of the left gastric artery, the region derives its blood
supply from above the diaphragm and has no mesentery. With hiatal transtraction,
the fundus is drawn through the hiatus and its vascular supply with it. Because
the fundus has a mesentery, the lesser omentum, the left gastric (coronary)
artery and vein, nerve fibers from the coeliac axis, peritoneum, fat, lymphatics,
lymph nodes and connective tissue are also crowded into the hiatus.
The gastric wall thickness is greater than that of the esophagus. Finally, the circumference of the fundus is many times that of the esophagus. This mass of tissue, with its greater cross sectional area, is pulled into a hiatus appropriate for a much smaller organ.
Some hiatuses are small
If these supporting tissues are drawn through the hiatus, with some patients
there may still be no obstruction because the hiatus is large or easily distended.
Reading surgical instructions for making the hiatus smaller, one might
suppose that the size of the hiatus is normally more than adequate. Because
there is an impression that hiatal size is a factor in HH, the surgeon may "correct"
a large hiatus with a few sutures.(43)
Some patients, however, have a hiatus that is small, barely large enough for
the esophagus itself, and not easily enlarged manually. Indeed, the hiatus itself
may itself be a cause of esophageal obstruction even without HH as in the following
patient:
CN SF-519A, male age 68. The patient states, "Meat and pancakes choke me up."
He has to interrupt meals to regurgitate food that won't go down. He has nocturnal
dyspnea and regurgitation of "foul tasting stuff" especially at night. Had an
excellent singing voice but lost it.
At fluoroscopy, barium passed freely through the esophagus. No HH could be
demonstrated nor was there any evidence of a LER despite the typical history.
The distal esophageal mucosal folds were coarse and reduced to three in number.
The deCarvalho test elicited gross reflux and, when it occurred, the patient
said that, in all but intensity, it duplicated his symptoms of acid regurgitation.
He managed to swallow two marshmallows each of which held up at the diaphragm
but was soon forced through by an effective peristaltic wave. On spot films,
the caliber of the lumen measured 1.8 cm across the momentarily impacted marshmallow
in the hiatus.
HH with a wide hiatus. Because of the wide hiatus, there is no hiatal constriction of the gastric blood supply, hence there is no swelling of the gastric mucosal folds above the diaphragm. Such patients do not get achalasia.
Compare with Fig. XIII.2
Surgeons calibrate hiatal size by the number of fingers that can be introduced
alongside the esophagus. Harrington(44) routinely
examined the hiatus in 1000 patients during upper abdominal explorations. He
found that in 55% of cases " . . . no opening could be felt around the esophagus,
35% admitted one finger, 8% two fingers and 2% three or more fingers."
[Emphasis added.] In operating on "short esophagus" HHs, H. Daintree Johnson(45)
" . . . was struck with the fact that . . . the hiatus often seemed within normal
limits."
When the gastric fundus and its mesentery are drawn through a small hiatus
by LMC, they exceed the available space so that the gastric lumen is compressed
from the start. Subsequent vascular engorgement from constriction of the vascular
and lymphatic return aggravates the obstruction.
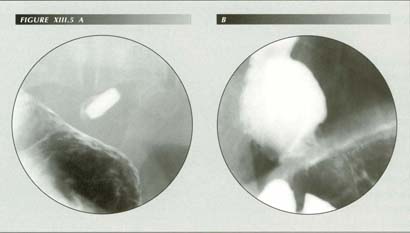
FIGURE XIII.5 A-B
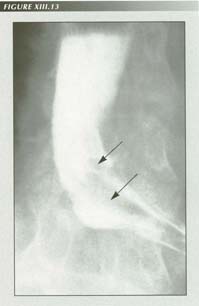
The hiatus itself may be a cause of dysphagia: A 2.5 mm barium tablet was arrested at the diaphragm (A). Compare tablet with the hiatal size in frame B. The patient also had an apparently normal, but fixed pylorus which neither contracted or expanded. His symptoms (�Feels like there is a clamp on my stomach.�) were completely relieved by endoscopic dilation of the pylorus.
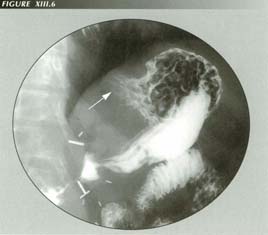
FIGURE XIII.6
Psuedo tumors of the fundus are reduced hiatus hernias: Enlarged, friable mucosal folds in the fundus can be mistaken for a neoplasm. these folds become swollen when the venous return from the fundus is impaired by hiatal constriction. They persist when it reduces.
There are many examples of true hernias in which strangulation occurs even
without the entire circumference of the organ herniating. In HH the entire circumference
is surrounded by a thin ring of potentially constricting diaphragm making strangulation
even more likely. There is, therefore, a priori reason to believe that
simple strangulation in the hiatus can occlude the lumen and produce the obstructive
appearance that is typical of the disease. Is there any clinical evidence this
is the actual mechanism?
Three cases
My experience bears out the strangulation mechanism. Casting aside preconceived
notions, most radiologists would, I think, concur that the appearance of DES
under the fluoroscope is absolutely typical of an obstructing bowel lesion.(47)
Because we have been taught to call it "diffuse spasm," we can totally overlook
the correct diagnosis. The following case is probably typical in that I mistakenly
believed I had done the job once I had identified a radiologic appearance with
a published description:
The patient was seen for severe dysphagia after he had lost 15 pounds in a few weeks and was unable to keep even keep water down. He had a ravenous appetite.
The fluoroscopic appearance of the esophagus was that seen with an acute mechanical
obstruction of the bowel. There was violent, rapid, churning esophageal peristalsis
that was almost completely ineffective in forcing barium below the diaphragm.
The forceful peristaltic waves lost control of the bolus as they approached
the diaphragm so that barium refluxed through the advancing ring of CM contraction.
The peristaltic wave immediately reformed and the process repeated. There was
a typical small tubular HH.
Pseudo tumor of the fundus: The venous return is compromised when a gastric segment is trapped above the diaphragm (A). The result is engorged and friable mucosa. Clinically, this accounts for the tendency of HHs to bleed. Such mucosa may bleed 1 unit/month without turning stools guiac positive. When the HH is again normally situated below the diaphragm (B), this engorged mucosa - and gastric wall as well- present as a tumor-like mass in the fundus. Provoking the HH (A) will cause the pseudo-tumor to vanish as shown here, but note the engorged folds.
I had described the appearance of an obstructed bowel without making the connection.
The esophagus is gut. Even after the successful therapeutic result
I was slow to realize that reducing the HH had relieved an obstruction.
One cannot always be operating from first principles. It is a useful shortcut
in everyday work to make a diagnosis when satisfied that published criteria
are met. There is also a time for proceeding from fundamentals, however, and
the earlier case was an alert when the following patient presented.
The patient was a man in his 20s who had the diagnosis of achalasia for several
years. A year previously he had a cardiomyotomy (Heller procedure) to treat
his dysphagia without obtaining significant improvement. Review of previous
films showed esophageal dilatation typical of achalasia but without redundancy.
On examination, a short tubular HH was demonstrated with extreme hyperperistalsis
that was ineffective in emptying the esophagus. The level of the obstruction
was at the diaphragm. The sphincter region was well above that level, but, because
the herniated segment of the stomach was the same diameter as the esophagus,
this could have been overlooked except that a slight LER identified the GE junction.
In addition there was now a pulsion type epiphrenic diverticulum in the distal
esophagus that had not been present before the myotomy.
With some trepidation, I made a diagnosis of hiatal strangulation but with
the caveat that this was a completely unorthodox diagnosis that went against
established opinion. Because the procedure dictated by conventional wisdom had
already been done, both patient and surgical consultant were willing to act
on that diagnosis.
Esophagoscopy on 10/15/68 revealed " . . . patent and voluminous reflux into the esophagus in the supine position. The terminal half of the esophagus was red, edematous and friable, but no distinct strictures were noted. There was a pulsion diverticulum of the lower lateral esophagus presumably at the site of his previous esophagomyotomy."
At a thoracotomy the following day, " . . . a left lateral esophageal diverticulum
was obvious and a small hiatus hernia was present. The normal anatomy of the
esophagus was reconstructed by involution of the pulsion diverticulum, and an
Allison type repair of the esophageal hiatus effected."(49)
Two weeks later the surgeon reported " . . . he is free of symptoms of reflux
and is able to swallow his food normally."
The third patient had a much longer period of observation. He had been followed
more than fifteen years for various GI symptoms with diagnoses of HH, severe
duodenitis, post-bulbar duodenal ulcer. There was no obstruction and no dilatation
at the last examination.
On reexamination in January of 1975, He had typical findings of achalasia with
delayed esophageal emptying, dilatation and redundancy of the organ. The experience
with the above two patients was described to the patient, his attending internist
and surgical consultants in another city where he elected to have treatment.
Preferring the conventional wisdom, he was treated by forceful dilatation and,
nevertheless, did reasonably well until his symptoms recurred 17 years later.
These cases illustrate several points: 1.) Simply reducing a hernia can relieve
the obstruction, 2.) It can relieve the obstruction even after the classical
Heller procedure has failed to do so. 3.) "Forceful dilatation" will also relieve
the obstruction. 4.) None of these clinical phenomena are consistent with the
postulated aganglionosis.
Cases of strangulated HH have been recognized. There are at least two reported
surgical cases(50)(51)
but these were seen purely as surgical emergencies and a connection with achalasia
was not suspected. Henderson(52) (Figures 16.3
and 16.4) illustrates a case of hiatus hernia with the manometric findings of
DES. Many other unrecognized cases can be found in monographs and the periodical
literature.
Boerema(53) reports(54)
that his operation, in which he pulls the stomach down and anchors it to the
posterior rectus sheath, although designed as a treatment for HH, gives excellent
results with both HH and achalasia! This is exactly what would be anticipated
if achalasia were a strangulated HH.
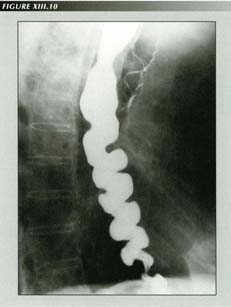
The significance of epiphrenic diverticula
There is additional evidence, from another quarter entirely, that HH can lead
to strangulation and obstruction. Because they are markers for obstructive disease,
it is no coincidence that epiphrenic diverticula (EPD) usually occur with either
HH, DES or achalasia. In cases of EPD, an incidence of HH of up to 50% is reported.(55)
Longitudinal studies of patients with such diverticula will show that they are
acquired and not congenital. They present the appearance of a "blowout" proximal
to an obstruction.
Epiprenic diverticulum for 34 years. These diverticula form as a buffer because, if there is obstruction at the hiatus, there is no other place for the bolus to go when peristalsis reaches the end of the esophagus. For the same reason, they are epiphrenic. Note the small hiatus. (B) the diverticulum is no longer in contact with the diaphragm because LMC has drawn 7 cm of stomach into a tube in an unsuccessful attempt to clear the obstruction. Note tenting of diaphragm. (T) Shortly after this film was made she was operated on for obstructive symptoms and the diverticulum resected! The result was satisfatcory, however, probably because the hiatus was widened incidentally.
Whatever the exact percentage may be, this association of uncommon diagnoses
cannot be dismissed as coincidental. It has prompted the naive conjecture that
epiphrenic diverticula may cause achalasia.(62)
Epiphrenic diverticula are typical pulsion diverticula. They consist of a
mucosal layer only. They arise because, propelled by the force of peristalsis,
esophageal contents find it easier to bulge through the wall of the esophagus
than to exit through the hiatal canal. It normally takes only 5 cm of water
pressure to force the sphincter from above. Obviously, it takes more force than
5 cm H2O to blow out the esophageal wall. The mere presence of EPD
therefore, is conclusive evidence of obstruction distal to its origin.(63)
As its name implies, the obstruction is at or very near the diaphragm. Fluid under pressure follows the path of least resistance. The reason for this is that early in the progression of a p-wave, the pressurized bolus merely distends the caudad esophagus. As it nears the end of its travel, there is no more esophagus to distend. The bolus must enter the stomach or, failing that because of a distal obstruction, either reflux through the p-wave or be forced through the esophageal wall. If the wall presents less resistance than the esophageal outlet, there must be a high grade obstruction of the esophagus outlet.
A diverticulum is not a totally useless structure to be extirpated at the earliest
opportunity. It serves a physiologic function as a buffer to contain a bolus
that cannot be forced through the usual channel by peristalsis. Without a buffer,
the incompressible liquid content of the esophagus must find an outlet when
compressed. The hypertrophied esophagus of DES may resemble a string of beads
because, when an en masse contraction of the circular muscle occurs,
alternate segments are either compressing the fluid or being blown out as buffers.
Such contractions are typical of esophageal obstruction.(64)(65)
Manometrically(66) they are distinguished by
the simultaneous rise in pressure at catheters placed at different levels.
Because epiphrenic diverticula are prima facie evidence of an obstruction
and occur with high frequency in achalasia, we can make a useful inference:
at some period in the genesis of achalasia, there was not only an obstruction
to esophageal emptying but also peristaltic activity that was forceful enough
to blow out the wall. But this is a description of diffuse spasm.
A toroidal Angelchik prosthesis closely mimics the toroidal appearance of gastric omentum in the PEL tent and thus the appearance of �achalasia�
From the above, in conformity with the mounting evidence in the literature
from the isolated case of Kramer et al. to the huge series of Vantrappen
et al., it seems reasonable to conclude that DES is the initial stage
of achalasia.
The obstruction is at the diaphragm
In achalasia, obstruction is not at the sphincter but at the level of the
hiatus. As the sphincter is 4-9 cm above the hiatus in patients with HH,
it might be thought that the observer should have no difficulty in establishing
this point. Yet it can be very difficult to detect the sphincter location in
HH with obstruction - especially if one is intent on demonstrating the "bird
beak." The fundus is drawn out into a tube that is easily mistaken for esophagus.
"Tramlines" similar to those seen in hypertrophic pyloric stenosis may extend
through the diaphragm and terminate at the effaced sphincter.
The point is important, because it exonerates the sphincter as the cause of
obstruction in HH. The sphincter may be as much as 8 cm above the hiatal
obstruction - a level at which one never sees narrowing in achalasia.
Degrees of hiatal obstruction
Only a few HH patients progress to achalasia, but those that do are the ones
with a small hiatus. Obstruction can range from partial to complete - from imperfect
clearing of the lumen by a peristaltic wave to life-threatening total occlusion.
Radiologically, several degrees of obstruction can be described. The earliest
stage is a failure of an effective peristaltic wave to clear the esophagus completely
with loss of control of the bolus.
A more pronounced degree of obstruction is the so-called "elevator esophagus."(67),(68)
After a barium swallow in the upright position, barium forms a persistent column
from the diaphragm to the middle or even upper third bounded above by an air-fluid
level. There is no organized peristaltic wave. Instead, en masse contractions
of the circular muscle occur as the esophagus attempts to propel barium distally.
The fluid level, instead of descending as the circular muscle contracts, rises
because the now narrower lumen must hold the same volume that a wider lumen
contained before the contraction. This produces an up and down "elevator" effect
on the fluid level. Tertiary contractions due to self-buffering may be seen.
Neither esophageal contraction nor hydrostatic pressure will clear the organ.
Often such patients will exhibit finely granular filling defects mixed with
the first swallow of barium. These represent mucus globules that have accumulated
overnight in the fasting patient. The first few swallows do not wash them out
of the esophagus because the hiatal squeeze is acting as a separatory funnel
to retain them while allowing barium to pass through.
Simultaneous or en masse contractions of the entire circular muscle
are characteristic of obstruction. They occur in various grades of achalasia
and have been produced experimentally in cats with an implanted inflatable esophageal
pneumatic cuff.(69) A Gore-Tex encircling band
produced simultaneous contraction in 85% of 17 cats so treated.(70)
When the band was removed in several animals, these abnormalities ceased. Kaye(71)
found no inconsistency between the manometric patterns and a "functional obstruction"
of the esophagus.
The most profound functional changes occur in patients with a severe obstruction. Here a dramatic churning peristalsis of the entire thoracic esophagus is seen. These patients may be in acute distress, unable ingest any food without vomiting and may have dramatic weight loss. In addition, a radiologist may note gagging, and bizarre tertiary contractions of the extreme degree sometimes described as "pseudo-diverticulosis" or "curling" of the esophagus. Again, one must search carefully to find the sphincter. It may be patulous and thus invisible without the clues mentioned. Such is the typical appearance of DES.(72)
In all three stages and in intermediate stages that could be defined, the obstruction
is at the diaphragm - not at the sphincter. When all else fails, finding the
sphincter is the key to the correct diagnosis. Perceptually, the TCs or "corkscrew"
or "pseudodiverticula" are the attention grabbers. The more subtle, but significant
findings locate the sphincter.
To summarize, we conclude that early stage achalasia is no more than the obstruction
that occurs with hiatal herniation. Achalasia and HH are never diagnosed simultaneously
because the "herniated" fundus is so tubular it is mistaken for esophagus.
Diverticula, true and pseudo, are buffers to which incompressible fluid is diverted
when the esophagus contracts vigorously to force an obstruction.
Some lessons from diffuse esophageal spasm
Hiatal obstruction of the fundus explains the entire picture of diffuse esophageal
spasm:
The churning peristalsis is the normal response of bowel attempting to force
an obstruction.
The "string of pearls" or "pseudodiverticulosis" appearance is due to self-buffering
of en masse esophageal contractions.
Corkscrew esophagus: Every book on the esophagus has at least one of these. This elderly gentleman had been followed for many years with frequent GI exams at leading institutions. The spectacular curling allowed the obstruction to go unnoticed. It is easy to see spiral muscle bundles in this case. The tracheal aspiration was asymptomatic and did not elicit a cough reflex.
Because hypertrophy is the normal response of bowel to chronic obstruction,
"idiopathic diffuse hypertrophy" is predictable, not idiopathic.
In the case of Kramer , the transition from DES to "achalasia" was not as rapid
as one would expect from the spectacular appearance of the violent peristaltic
and non-peristaltic contractions. Their patient was followed for eight years
as he developed increasing symptoms of obstruction with fifteen manometric or
balloon kymographic studies. They reported that " . . . symptoms, oesophageal
radiographs, balloon kymographic and manometric records were diagnostic of diffuse
spasm." After forceful dilatation the same studies " . . . were quite characteristic
of cardiospasm." Although this was not a happy result, in other cases,(73)
complete restitutio ad integrum occurs.
The hypersensitivity to Mecholyl reported by Kramer and Ingelfinger(74) was present in this patient both when he was considered to have "diffuse spasm" and when he exhibited the picture of cardiospasm. Barrett also noted a transition from DES to achalasia. Kramer et al.(75) had earlier suspected that diffuse spasm and cardiospasm might be related because some but not all patients with DES also have a positive Mecholyl test.(76)(77)
Enlarging the hiatus relieves both DES and achalasia
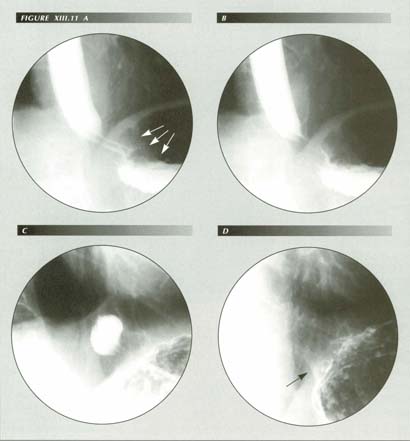
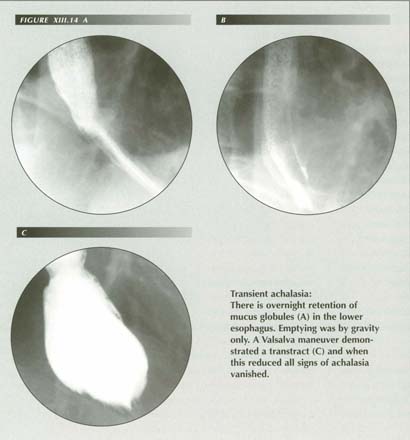
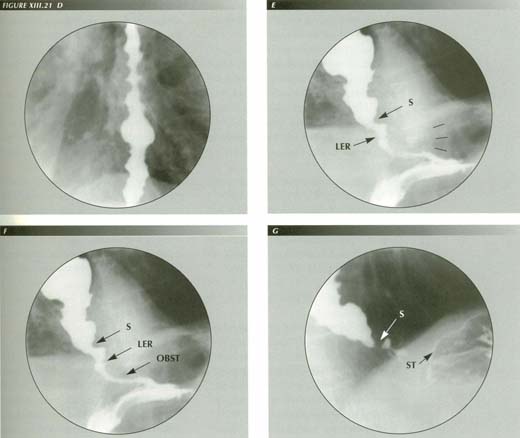
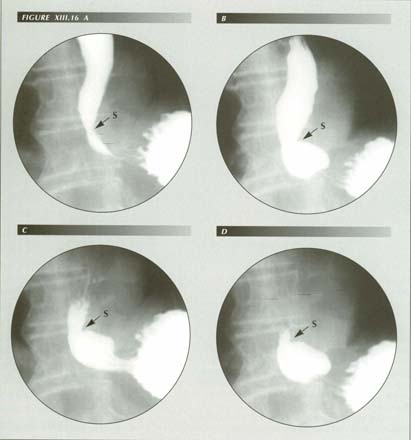
Achalasia can clear in seconds: Frame (A) shows an esophagus emptying slowly by hydrostaic pressure alone as there is no peristalsis. Note the �tramlines� similar to those seen in hypertrophic pyloric stenosis passing through the hiatus and the �bird beak� termination at the diaphragm. The latter appears to be partly surrounded by a 3 cm soft tissue mass in the gastric air bubble. The beak of the �bird� tends to approach the horizontal because the esophagus is redundant because of the 4.5 cm HT that is not seen until frame (C). Later frames showed normal peristalsis with excellent peristaltic cleanup despite a grade ii esophagitis. The soft tissue mass has vanished! Frame (D) shows the subtle, yet unmistakable edge of ta trumpet which indicates powerful LMC that has released the trapped fundus by reducing hiatal squeeze.
In dilating the sphincter, however, the operator is unwittingly dilating the hiatus because the hourglass shaped dilating bag self-centers, not on the sphincter, but on the hiatus. As another instance of the Law of Compensating Errors, things turn out right for the wrong reason. Relief of the hiatal obstruction follows enlargement of the constricting ring.(79) As with achalasia, dilatation restores the normal peristaltic wave in DES.(80)
It should be noted that the force used to dilate the "sphincter" is completely
out of proportion to the delicacy of the structure being dilated. Anatomically,
the sphincter is so tenuous that only recently have anatomists been willing
to grant its existence. The esophageal hiatus, unequivocally anatomical, is
more likely to be offering the resistance to distention.
Such therapeutic results are inconsistent with either an aganglionosis or
a "disordered motor function." That an almost brutal stretching procedure could
reverse a degenerative process or reorder an incoordination of motor function
is inconceivable. On the other hand, dilating an obstruction can be expected
to cause a return to a normal peristaltic mode.
Self-limited hiatal strangulations
It could well be objected that if 55% of patients have a hiatus that will not
accommodate even one finger, there should be a much higher incidence of strangulation
than is encountered, given the very high incidence of HH. This objection is
valid, however, it is not really an objection. It merely shows that lesser degrees
of strangulation usually go unrecognized.
There are many cases of temporary or intermittent strangulation of the fundus
in the hiatus that reduce spontaneously. The portion of the fundus above the
diaphragm becomes edematous and engorged. Seen after it has reduced, this engorgement
may easily be misinterpreted as a "fundic gastritis" or, with potentially disastrous
consequences, as a neoplasm, because the fundus appears separated from the diaphragm.
The appearance is so diagnostic - a fundic "mass" concentric with the esophageal
orifice - that one can make the diagnosis of "hiatus hernia without herniation."
Mucosal engorgement of the retracted portion of the fundus is so common it
is an endoscopic sign of HH. Endoscopists " . . . frequently find friable or
hemorrhagic gastric mucosa in the presence of normal esophageal mucosa in patients
with symptomatic hiatus hernias."(81) Morrisey(82)
notes " . . . relatively little attention has been paid to the gastric mucosa
just distal to the mucosal junction in patients with reflux esophagitis. This
mucosa often appears erythematous, friable and occasionally frankly eroded."
A biopsy of the friable mucosa may be reported as normal because there are no
inflammatory cells. This and the finding that some hiatuses hug the scope tightly
are significant in elucidating the mechanism of strangulation. The mucosal changes
affect only the organ with the constricted blood supply, that is, the portion
of the fundus above the hiatal constriction. The esophagus, with an unimpaired
blood supply, may appear normal to the endoscopist.
I have occasionally seen hiatal obstruction with spontaneous reduction and remission during a fluoroscopic examination:
DW 46873 M393. Female, age 52 Fluoroscopic note: Ingested barium showed moderate
dilatation of the esophagus and a delay in emptying. A fluid level formed at
the height of the aortic arch and only slowly descended. Unorganized, irregular
contractions of the esophageal outline were noted. They would appear on one
side without deforming the opposite side as may be seen with partial LM contractions.
They produced no effect as far as emptying the esophagus. The dilatation was
so pronounced that the left atrial border indented the esophageal outline sharply
enough to at first suggest enlargement.
Although these findings were all typical of achalasia, when she performed a prolonged Valsalva test a typical HH with a lower esophageal ring appeared. As this reduced, all signs of achalasia disappeared, an organized peristaltic wave formed and the esophagus emptied completely.
HHs frequently get stuck above the diaphragm temporarily and then reduce when
the stomach distends. This explains their bleeding propensity even without esophagitis.
All lesions in the cardia have a greater tendency to bleed than when found elsewhere
in the esophagus or stomach - a further indication of the strangulating effect
of even a large esophageal hiatus.
The "inflammatory" gastroesophageal polyp: a minimal strangulation
�Inflammatory� gastro- esophageal polyp: the term is probably a misnomer. Note the tight hiatus which constricts circulation in the portion of the stomach retracted above the diaphragm. This is a minor degree of pseudotumor of the fundus.
Although up to 1984 there were reports of only eleven cases,(83)
the condition is common enough that many examples can be collected by the interested
observer. Its frequent appearance is testimony that in many patients the hiatus
is barely large enough to hold the esophagus because the appearance can only
be understood in the light of the local blood supply. This is the only region
of the esophagus that receives a blood supply from below the diaphragm. Impairment
of the venous return produces local vascular engorgement. The existence of the
fold is proof that incarceration of the retracted fundus is not momentary or
infrequent.
Other hiatal obstructions look like achalasia
The conditions that "mimic" achalasia have great significance. Surgical operations
about the hiatus including a too tight HH repair, post-vagotomy periesophagitis(84)
and, particularly, a tight fundoplication(85)
can simulate the appearance including the "bird-beak," esophageal dilatation
and "motor disorders" (repetitive and simultaneous contractions). In one way
or another, these procedures all constrict the tissue mass in the hiatus.
Kumar reported a cartilaginous esophageal ring similar to a tracheal cartilage
in the esophagus.(86) It was indistinguishable
radiologically from achalasia. The abnormality cleared after resection.
Tumors that invade the hiatus provide a more direct, unequivocal reproduction
of achalasia. In the following case a tumor of the hiatus, situated half above
and half below the diaphragm produced the picture of achalasia.
LL 6872 7/6/64. Female, age 76. At fluoroscopy barium passed freely through
the hypopharynx but arrested at the diaphragm. At this point the esophagus showed
a long taper to a caliber of a few millimeters. This portion of the esophagus
pulsated violently from transmitted cardiac contraction. In the upright position,
a fluid level formed above the aortic arch and fluid trickled only slowly through
the esophagus. A peristaltic wave would begin to form below the aortic knob,
but this never progressed distally. On one or two occasions there was some reflux
through the advancing peristaltic wave into the proximal third of the esophagus.
The esophagus was only slightly dilated. The cardia was narrow and somewhat
separated from the diaphragm.
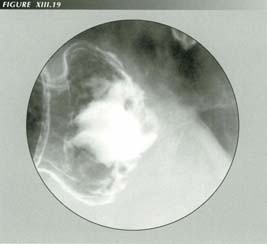
A typical �bird beak� of achalasia. The �gastroesophageal polyp� (arrows) identifies stomach above the narrowing.
"Secondary achalasia"
It is well known that the radiologic differential diagnosis of achalasia from
carcinoma of the fundus of the stomach is impossible. (87)"Idiopathic"
and "secondary" achalasia are identical clinically, radiologically, manometrically
and endoscopically. Even the Mecholyl test(88)
and Seidlitz powder tests do not distinguish between achalasia and these conditions.(89)(90)
This has led to the notion of secondary achalasia - that is, achalasia
to which a cause can be assigned. In the three cases of secondary achalasia
encountered by Ferguson and Burford,(91) even
the endoscopic appearance was the same as in primary or idiopathic achalasia.
In the seven cases of regional carcinoma reviewed by Tucker, Snape and Cohen(92)
the manometric profile was identical whether the carcinoma was of the stomach,
the lung or the pancreas. The endoscopic appearance suggested carcinoma in only
two cases. They concluded " . . . the radiographic, endoscopic and manometric
studies . . . may not discriminate between the primary and secondary forms of
achalasia." Sandler and associates(93) also
found clinical criteria unable to distinguish the two.
Such cases pose insuperable problems for the aganglionosis theory of achalasia.
To preserve the theory, speculation now must center on how carcinoma invading
the myenteric plexus simulates or causes an aganglionosis. When searched for,
however, such plexus invasion has not been found.(94),(95)
A carcinoma-induced peripheral neuropathy has been suggested. This might preserve
the aganglionic theory, but it is hard to conceive of a neuropathy limited to
the esophagus. Nor is it reasonable to believe that five different types of
carcinoma could all produce an identical, highly selective neurotoxin.
There is a simpler explanation: there is no difference between primary and secondary achalasia. All cases are secondary to hiatal obstruction. Retraction and incarceration of the fundus produce hiatal obstruction just as surely as does carcinoma invading the hiatus.
Again, Occam's razor dictates favoring the explanation that requires the fewer assumptions. A tumor invading a passageway will obstruct it. That is enough to explain the appearances. When the lesion is local, why make the additional assumption of involvement of a plexus that extends the entire length of the esophagus to explain the appearances? Moreover, esophageal carcinoma arising above the hiatus also invades the myenteric plexus but never causes the appearance of achalasia even if it obstructs.
Achalasia clears when hiatal tumors regress.
A single case report of Davis et al.(96)
is of great interest because it illustrates both the ability of a hiatal tumor
to produce achalasia and that achalasia resolves with tumor regression. This
patient had both generalized reticulum cell sarcoma and typical manifestations
of achalasia including marked obstruction and uncoordinated contractions. Manometric
studies were reported as typical of achalasia. A radioactive gallium scintiscan
showed a concentration of activity from D-10 to L-1 in approximately the midline,
anterior to the vertebral column [including the hiatal area] and posterior to
the liver. His esophagus was restored to normal functionally and radiologically
after two weeks treatment with prednisone and vincristine.
The scans before and after treatment prove hiatal obstruction and relief. It could be objected that the hiatal area was never explored surgically for confirmation. This objection does not apply to Kline's(97) case of "vigorous achalasia" diagnosed by manometric findings of increased sphincter pressure (30-40 mm Hg) and simultaneous repetitive contractions. Endoscopy suggested achalasia and the radiological findings were those of dilatation of the body and narrowing at the GE junction. On exploration, an anaplastic gastric lymphoma was found extending from the GE junction to mid-stomach. Manometric and radiologic findings reverted to normal one month following treatment with cytoxin, vincristine, adriomycin and prednisone.
Nelson and Horsley's case(98) of idiopathic
retroperitoneal fibrosis produced the typical long, beak-like narrowing centered
on the diaphragm typical of achalasia. At exploration, a fibrotic mass arising
on the posterior peritoneum enveloped the great vessels, kidneys and both crura
of the diaphragm. It extended 2 cm into the mediastinum " . . . enshrouding
the esophagus at the GE junction." After it was dissected out, the patient had
complete relief of her dysphagia.
In the above report, the authors concluded that " . . . neoplastic compression
or infiltration of the esophageal myenteric plexus produced the pseudoachalasia."
An aganglionosis, however, should be irreversible - degenerated nerve tissue
does not un-degenerate. Regression of nodes or infiltrate obstructing the hiatus,
on the other hand, relieves the obstruction. The patient is not wrong ("pseudo
achalasia"); the theory is. Nevertheless, pseudo achalasia has persisted as
a diagnostic entity.(99)
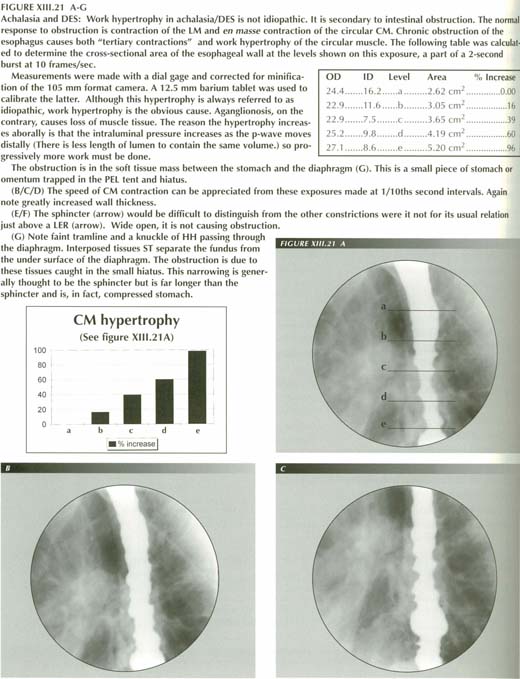
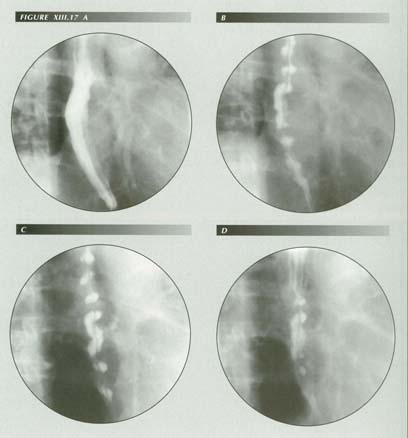
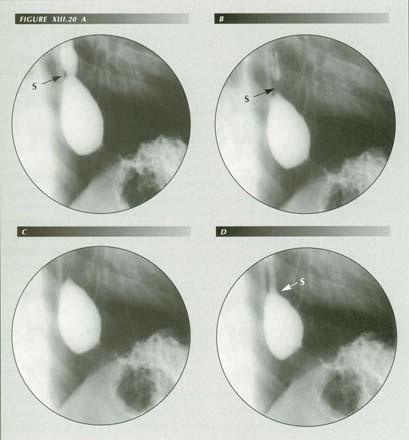
A slightly more severe stage of achalasia: The nearly horizontal �bird-beak� configuaraion (frames A&B) and tramline shadows (frames B-D) are shown to be in the transtracted stomach by the fact that the p-wave -- which stops at the sphincter -- ends well above the diaphragmatic constriction. A good clean-wiping p-wave is still able to force the partial obstruction (C & D). During this stage, the CM will undergo work hypertrophy
These tumors and other disorders (amyloidosis(101),(102)
and post-vagotomy periesophagitis(103) can
be added to the list) do not mimic achalasia. They are achalasia -
indistinguishable radiologically, manometrically, pharmacologically and, usually,
endoscopically from the more usual cause of hiatal obstruction of the gut.
The esophagus can also be obstructed by tightly wrapping the fundus about it
as in a Nissen fundoplication. Such obstruction affected 9 of the 38 fundoplications
requiring reoperation by Leonardi and Ellis.(104)
They either manifested achalasia (7) or diffuse spasm (2).
Tertiary contractions: An esophagus without a functional p-wave will contract en masse. This is the fallback mode when obstruction is encountered. Tertiary contractions are a complex manifestation of a.) LM shortening. [Note the losss of the posterior bowing of the esophagus as the TC�s form.] b.) Mass circular muscle contration. c.) Self buffering -- an en masse contraction of circular muscle cannot compresss the liquid contents of the organ. The stronger muscle bundles overpower the weaker resulting in alternate constricted and widened segments. Modern anatomical research has shown that the muscle of the body is in part spirally arranged, a fact that is well shown during such contractions. There appear to be two spirals -- one ascending from below, the other descending from above. The velocity of both LM and CM contraction is remarkable as can be seen on these exposures -- 1/10th seconds apart
It is more difficult to come to grips with the "nonspecific esophageal motor disorder" concept that now envelopes the DES-achalasia spectrum because its very vagueness protects it. I suppose it to mean that in some fashion, the central or peripheral program controlling the end organ has developed defects that destroy coordinated muscular function. That 4 mg of vincristine, stretching the hiatus or a surgical procedure could reprogram the controller is difficult to conceive.
What all treatments have in common is that they relieve an obstruction. When
they do so, the "disordered motor function," that is not disordered at all but
merely the normal reaction of gut trying to force its contents through an obstruction
- improves as the esophagus reverts to normal function just as would the small
bowel after lysis of an adhesion.
The "bird beak" of achalasia has no resemblance to the sphincter.
The radiologic appearance of achalasia is not that of the physiologic sphincter.
The typical beak-like termination of achalasia bears no resemblance to the LES.
It is not only too aboral, but is also too long - 3 cm or even 4 cm - whereas
the LES is scarcely over 1 cm in length.(106)
The characteristic feature of the "beak" - its biconcave outline - is due to
the torus of mesentery in the phrenoesophageal tent that surrounds and constricts
the herniated fundus. Also typical of a squeeze effect is the "tramline" or
"twin track" appearance due to barium on either side of an enlarged mucosal
fold. It is virtually identical with the tramlines due to the mucosal squeeze
of hypertrophic pylorus stenosis.
Advanced achalasia: (A) Dilated, redundant esophagus. (B) Narrowing just behind but above the dome of the diaphragm.
Denervation is a result, not a cause of achalasia.
Under the conditions prevailing in advanced achalasia, many muscle cells are
effectively denervated. Stretching neurons and their processes in two directions
to many times their normal length may exceed their elastic limit or even avulse
them from motor end organs. The terminal varicosities of the axons protrude
through ultramicroscopic holes in an enveloping glial sheath to deposit their
neurotransmitter in the connective tissue near the smooth muscle cells.(107)
Overdistending the esophagus should damage these contacts.
The peristaltic gap
In deglutition, LMC normally tents the distal esophagus and adjacent fundus
so that the bolus drops into the stomach when the sphincter opens. When LMC
elevates a tubular segment of fundus above the hiatus producing a hiatal squeeze,
it causes a fundamental disorder of propulsion. The esophagus cannot hand off
the bolus to the stomach in the normal way. As we learned when analyzing the
captive bolus, esophageal peristalsis stops at the sphincter.
Fundic peristalsis: The most proximal gastric p-wave I have ever encountered was in this severely nauseated patient. Gastric peristalsis cannot take over when the esophageal p-wave stops.
An aperistaltic segment forms as surely as if the bowel had infarcted or been
stripped of its ganglion cells. As elsewhere, intestinal obstruction is the
result of such a peristaltic gap. So, in this sense, the appearance is the same
as though it were a true aganglionosis. It is a failure of peristalsis,
not an aganglionic segment, that produces the characteristic signs of obstruction.
Aperistalsis produces an "obstruction" even without an occlusion - thus accounting
for otherwise typical cases of "achalasia" with complete sphincter relaxation.(109)
This situation is unique in the alimentary tract. It may account for the ease
with which achalasia has long masqueraded as an aganglionosis. It also explains
why the compensatory work hypertrophy that results from hiatal obstruction stops
at the sphincter well above the actual obstruction at the diaphragm.
As a result, there are two reasons for obstruction: the hiatal squeeze
and the aperistaltic segment. In practice, the hiatal squeeze need not even
be very tight: the peristaltic gap alone is enough to account for the appearances.
Strictly speaking, the sphincter has nothing do with the case. Its tone and its ability to relax are unimpaired. They do not enter the equation as a third cause of obstruction. In that sense the emphasis placed on sphincter tone by conventional wisdom is misplaced. In another sense, however, the sphincter is all-important.
Sphincter latching
It will be recalled from the description of the captive bolus phenomenon that the advancing p-wave "latches" the LM. The more distal the wave, the shorter the esophagus. From this it was concluded that the LM, like the CM, is activated by the p-wave passing through it but, unlike the CM, the LM does not relax in the wake of the p-wave.
Although it contracts incrementally, the LM relaxes all at once. Progression
of the p-wave into the sphincter is the signal for LM relaxation. Then roles
are reversed. Now it is the CM of the sphincter that does not relax
in the wake of the p-wave. The p-wave has stopped. Having fulfilled its function
of opening the sphincter in the earlier stages of deglutition, the LM can now
relax. The antireflux role of the advancing p-wave transfers to the sphincter
that now latches in the closed position.
Failure of sphincter latching: this patient with chronic reflux had a mild impairment of the p-wave. The feeble p-wave cannot latch the sphincter and, on encountering back-pressure because of the temporarily occluded PEL tent, gives way and allows reflux back into the esophageal body (D). This may be an important factor in the muscle hypertrophy of achalasia.
The unphysiologic conditions of fundic strangulation interfere with the ability
of the sphincter to latch. In DES, the early stage of the disease, one sees
very forceful p-waves that reach the lower esophageal sphincter only to lose
their grip on the bolus. The bolus refluxes into the body of the esophagus stimulating
another secondary p-wave. This would not happen if the sphincter were latching.
This failure may be due to increased pressure below the sphincter. Normally, the infrasphincter region is exposed to intragastric pressure at the instant of sphincter latching. In fundic strangulation, this pressure - the full pressure generated by the p-wave - is much greater. Mittal and his associates(110) have shown that esophageal clearance of refluxed acid is not a normal stepwise increase in pH if the subject has a HH. Clearance then becomes biphasic because each swallow induces a new episode of reflux. This would not happen if the sphincter stayed latched until the next p-wave formed.
A summary of the evidence
At this point, it would be well to summarize the major items of evidence that
hiatal obstruction is the sole cause of diffuse esophageal spasm and
achalasia.
Hiatal occlusion by tumor or fibrosis produces a radiologic, endoscopic, pharmacologic
and manometric picture indistinguishable from DES or achalasia.
Removing a tumor obstructing the hiatus by surgery or chemotherapy causes
regression of EMDs and achalasia.
The frequency of the sequelae of hiatal strangulation - "pseudotumor of the
fundus" and "inflammatory gastro-esophageal polyp" shows that strangulation
is a common event.
In many patients the hiatus is not easily distended but is just adequate to
allow passage of the esophagus .
Remission of DES occurs after surgical reduction of a strangulated hiatus
hernia as in the two cases described here. Boerema reports other cases of achalasia
relieved by simple gastropexy.
The high incidence of epiphrenic diverticula with HH, DES and achalasia proves
distal mechanical obstruction in all three.
The fluoroscopic appearance of DES is that of acute mechanical obstruction
of the bowel.
Hypertrophy of smooth muscle is characteristic of intestinal obstruction.
An aganglonosis should cause atrophy.
Longitudinal studies show a transition from DES to achalasia.
The radiologic appearance of the distal esophagus is not that of a tight but
otherwise normal sphincter. It is the appearance seen in the captive bolus test
when the stomach is obstructed in the PEL tent by mesentery crowded into it.
No test will distinguish esophageal obstruction due to tumor from obstruction
due to strangulation.
Mechanical dilitation may relieve the symptoms but dilates the hiatus, not
the sphincter.
Whereas it is physiologically impossible for degeneration of a neuron network
to cause hypertrophy of the muscle it supplies, work hypertrophy is a normal
reaction to obstruction of the gut.
This evidence - most of it inconsistent with either the aganglionic hypothesis or the more nebulous EMD formulation - shows that acute and chronic hiatal obstruction, most commonly by fundic strangulation, are the respective causes of DES and achalasia.
The evolution of "diffuse muscular hypertrophy"
The failure of sphincter latching and the peristaltic gap are the keys to
understanding why obstruction at the hiatus causes hypertrophy and/or dilatation
of the proximal esophagus whereas benign strictures and carcinoma do not. This
distinction has been a last refuge of the aganglionosis hypothesis.(111)
The evolution of the full-blown picture of achalasia from the appearance of
esophageal strangulation can be reconstructed. The forceful peristalsis of DES
may succeed in reducing the strangulation or it may not. In the former and most
common case, achalasia does not occur but the engorged mucosa on reduction may
present the appearance of a fundic gastritis or pseudotumor.(112)
In the latter case, hyperperistalsis and compensatory hypertrophy of the circular
muscle may still force the obstruction. This leads to the circular muscle thickening
found by the surgeon at cardiomyotomy(113)
or, in the extreme case, to the "diffuse muscular hypertrophy" described by
Johnstone(114) and others(115),(116),(117),(118)
If one reads the description of these cases carefully and examines the illustrations,
in those cases in which a determination can be made, a HH was present. Some
are mentioned in the report, but some, although clearly shown in the illustrations,
were not reported by the radiologist because the fundic transtract was the same
diameter as the esophagus.
A tubular fundus is easily mistaken for esophagus even at thoracotomy. Why
else would the operative report say " . . . the lower esophagus was greatly
thickened except in the terminal 4 centimeters where it was normal."
[Johnstone, Case I - also Sloper's Case 5] or "The cardia and distal 3 cm
of the esophagus were normal but immediately above this segment the esophageal
wall was thickened beyond 1 cm and felt like a sausage." [Johnstone, Case 3].
(Emphasis added.) Sloper's Case 4 and a case of Rake's also describe the same
phenomenon.
The fundus and the esophagus distal to the sphincter do not undergo work hypertrophy
because, being aperistaltic, they do negligible work. The p-wave stops at the
sphincter. There is no conceivable reason hypertrophy would stop short of the
sphincter. We conclude that, even in these surgically explored cases, the fundus,
drawn through a small die-like hiatus, was mistaken for esophagus.(119)
In addition, the radiologic picture is in no way different from DES. The preponderance
of the hypertrophy, as would be expected in work hypertrophy, is always
in the circular muscle layer whenever the two layers are described separately.
In Sloper's illustrations, instead of being somewhat thinner than the LM layer,
the CM is over twice as thick.
The radiological and manometric signs of hyperperistalsis and en masse contractions, perforation in one case and an epiphrenic diverticulum in another complete the proof that all of Johnstone's examples of "idiopathic diffuse muscle hypertrophy" were due to normal work hypertrophy from hyperperistalsis secondary to strangulated HHs.(120) Again, assuming an "idiopathic" etiology is redundant when obstruction, the usual cause of muscular hypertrophy, is patently present.
A critical point in the evolution of achalasia occurs when the dilatation becomes sufficiently extreme and/or the circular muscle becomes sufficiently exhausted that the peristaltic wave can no longer approximate the esophageal walls sufficiently to obliterate the esophageal lumen.
End-stage exhaustion
Unless the lumen can be obliterated, peristalsis has no propulsive force.
Somehow sensing the futility of peristalsis, the organ compensates by a pattern
of en masse contraction that does have a propulsive force. Counterproductively,
this is often dissipated by self buffering presenting as tertiary contractions.
After that, dilatation and elongation are the only responses of the esophageal
wall to the ingestion of food as the organ degrades to a passive conduit. Hydrostatic
pressure of the fluid column, perhaps with some assist from a Valsalva maneuver,
is the only force that can even partially empty the esophagus. This, of course,
is the usual presenting appearance of achalasia. Except for the unique position
of the esophagus as the intrathoracic gut, the appearance does not differ from
chronic intestinal obstruction elsewhere in the alimentary tract.
Qualman et al.(121) after reviewing
the pathologic literature noted that "The neuropathologic findings generally
reported in [achalasia] include chronic inflammatory infiltrates within the
esophageal myenteric plexus and degenerative changes within smooth muscle or
nerve fibers." The hypersensitivity to cholinergic drugs is lost when the end
organ fails. Ganglion cell degeneration is a result, not a cause of the obstruction.
One does occasionally see indentations of the outline of the decompensated
esophagus as though the circular muscle were contracting erratically. These
indentations, however, cannot be due to circular muscle contraction because
they occur on only one side.
The contribution of manometry to the confusion
It now appears(122),(123)
that, given the proper transducer, the term "cardiospasm," although also incorrect,
need never have been replaced! Measurements with the old style non-perfused
catheters showed no increase in LESP. However, measured with the newer perfused
catheters,(124) LESP is twice the normal pressure.
Measured with the still newer intracorporeal transducers LESP is sometimes normal.(125)
Paradoxically, Katz, Richter, Cowan and Castell(126)
found that 30% of their patients with otherwise typical achalasia had complete
relaxation of the sphincter. They conclude, "Apparent complete LES
relaxation may be seen during manometry in achalasia and should not exclude
its diagnosis." Anachalasic achalasia seems the final reductio ad absurdum
of the aganglionic hypothesis. The detailed mechanics of the disorder, as I
have shown, make these inconsistent measurements of LESP/hiatal squeeze understandable.
Although manometry was a technical refinement, it started and perpetuated the confusion. Despite the changing physics of pressure sensing equipment that caused embarrassing reversals when absolute pressure measurements were involved, it is beyond doubt that relative pressure measurements and wave patterns are meaningful. However, in DES and achalasia, these patterns do not represent a spectrum of intrinsic neuromuscular disorders, they are the normal motor response of a gut trying to overcome an obstruction that is in part mechanical and in part interrupted peristalsis, Initially, this response is unusually forceful and unusually frequent peristalsis. Later en masse contractions occur. In the end, hypertrophy or exhaustion and decompensation occur.
The hiatal ring can be very resistant to expansion.
Thus far, I have tacitly assumed that the hiatus has enough intrinsic strength
that it will not easily stretch to accommodate a herniated fundus. The diaphragm
is a thin structure. It contains considerable muscle, the fibers of which are
easily stretched. Even if the fundus were retracted into the hiatus, it could
conceivably act as its own dilator, expanding the hiatus and so forestalling
strangulation or entrapment. It is natural to ask, "Does the hiatal ring have
enough strength to be a persistent strangulating constriction?"
Unless one is a surgeon, he has no opportunity to explore the hiatus personally
to see how well it can resist stretching. Fortunately, we have exact information
about the resistance of the hiatal ring: the current treatment for EMDs is balloon
dilatation. Up to 740 mm Hg of pressure is applied to the hiatus (under the
impression it is the sphincter) and even this may not be enough to dilate it
adequately. Comparing this with the 150 mm Hg used to inflate a blood pressure
cuff gives an idea of the force generated by that pressure. In some cases even
this force is insufficient to produce enough hiatal dilatation to relieve the
symptoms and the procedure may have to be repeated three or four times to stretch
the "sphincter" to a diameter of as much as 5 cm.(127)
Why haven't surgeons found the strangulation?
The wonder is that a strangulation or hiatus hernia is not recognized at the
time of the operation. The skeptic is thinking, "Logic is well and good, but
surgeons can see what is going on. They have been looking at these
cases for a hundred years. They can't all be missing it."
In my original case, the surgeon who performed a Heller procedure was not
alerted what to expect and did the operation without comment and without reducing
a HH. The second surgeon, with whom the rationale of the patient's problem had
been discussed, did see the hiatus hernia, reduced it and cured the patient
although in repairing the blowout he was undoing the myotomy. We have seen that
in Johnstone's cases the only explanation of hypertrophy stopping short of the
diaphragm was a tubular HH not appreciated by the operators.
A Heller procedure is usually done in an advanced stage of the disease. It
appears from illustrations that there is a marked hypertrophy of the circular
muscle. It may have taken years to develop this "musclebound" condition that
it can be constricting. The original strangulation may have long subsided.
Good results are reported no matter what operation is done. This may not be
mere surgical self-congratulation. By the time the area of interest is exposed,
landmarks identified, etc., the strangulation may be inadvertently reduced.
If the procedure is done by the abdominal approach, and this is preferred by
many,(128) any HH present will be reduced when
pulling on the stomach to get at the esophagus.
Done by the transthoracic approach, a partial fundoplication (that by necessity
gets the fundus out of the hiatus thus relieving the strangulation) is regarded
as one of the three principles necessary for success.(129)
In the case of the Boerema "anterior gastropexy" of course, the good results
are due to pulling the stomach down forcefully and so reducing the HH.
Typically the surgeon finds what he expects to find - a thickened segment
of the esophagus similar to a hypertrophic pylorus. Doing a myotomy on this
hypertrophied muscle, could be effective treatment accidentally. Any incarcerated
fundus will be released when the incision is " . . . carried down into the circular
fibers of the cardia." which would be impossible without either dividing the
hiatal ring or recognizing that the fundus was already above the hiatus.
Review of a large number of surgical reports turns up the reassuring fact
that often the hiatus hernia is not missed at surgery. Most Heller
procedures are "modified" and the modification almost invariably involves repairing
a HH unsuspected preoperatively or adding an antireflux procedure to forestall
a frequent complication of the Heller procedure.(130),(131),(132)
This may include extending the myotomy into the stomach. Despite their use of
a transthoracic approach, Ferguson and Burford, for example,(133)
recommended opening the diaphragm to detect HH after their experience of finding
three HHs(134) unsuspected radiographically
and at esophagoscopy and three cases of adenocarcinoma of the fundus that produced
" . . . symptoms, esophagoscopic, and x-ray changes indistinguishable from achalasia."
Significantly, they also report that most of their unsatisfactory results were
due to hiatus hernias either missed at the time of myotomy or possibly
caused by " . . . surgical manipulations about the GE junction."
Ellis et al.,(135) for example, report
that 16% of their cases had already had an esophagomyotomy and another 20% had
HH or a "lax hiatus" at the time of exploration. Several others had actually
had a HH repair when originally operated for achalasia!
The actual situation at the time of the procedure may not be as straightforward
as the neat anatomical drawings suggest. There may be an additional 20% of strangulations
missed as that is about the incidence of unsatisfactory results with surgery.
Nissen, Belsey, Collis and Boerema procedures may be done concurrently with
the myotomy.(136)
Trounce et al.(137) found that when
the narrowed segment as determined by cine-radiography [i.e., a tubular fundic
transtract] was examined at operation, " . . . its muscular walls appear quite
normal, in notable contrast with the hypertrophied and dilated esophagus above."
Johnstone's cases 1 and 3 were similar suggesting these observers mistook a
tubular section of stomach for esophagus. Peristalsis ends at the sphincter
and so does hypertrophy. These are highly experienced experts on the area and
they all saw the hiatal transtract and commented on it in their articles but
had no reason to believe was anything but a segment of normal esophagus. The
resemblance to esophagus must be striking, indeed.
It is also possible that the diagnosis may be overlooked at surgery because it takes very little tissue to produce obstruction in a small hiatus. It will be recalled that Harrington found that 55% of patients have a hiatus too small to admit one finger. When a tag of fundus or fat further occludes it, it may not be obvious to a surgeon whose attention is directed primarily, not to the hiatus, but to the organ he is operating upon. The truism that we only see what we are looking for probably holds true for surgeons as well as others.
Therapeutic implications
Boerema(138) fortuitously discovered that
a pulldown procedure intended to correct a HH alleviated cardiospasm. It would
be better to do the procedure in the early stages of strangulation instead of
after the organ decompensates. Fundoplication and "snugging" the hiatus during
operations for hiatus hernia or "restructuring the cardia" are irrational and
should be abandoned. If anything, the hiatus should be enlarged so
that a recurrence will not aggravate the original difficulty.
The Heller muscle-splitting procedure seems to make no sense at all. No one would dream of treating compensatory hypertrophy of the small bowel proximal to an adhesion in this fashion.
SUMMARY
Although solidly entrenched for over sixty years, the aganglionic theory of
achalasia topples in a welter of contradictions, unverified consequences and
unjustified assumptions. The notion that a neurologic deficit can explain muscle
hypertrophy is nonsense.
Like most esophageal disorders, both diffuse esophageal spasm and achalasia
can be traced to abnormal function of the longitudinal muscle. A forceful contraction
of the LM, such as that occurring with pyrosis, nausea, gagging or vomiting,
produces a "hiatus hernia" in which the fundus, epiphrenic fat, lesser omentum
and the left gastric artery or its branches are drawn up through a small
hiatus where they become incarcerated.
The incarceration may subsequently reduce spontaneously, in which case there
will be a "fundic gastritis" or a "pseudotumor of the cardia" caused by the
vascular engorgement of the formerly incarcerated fundus. If it does not reduce,
the appearance of "diffuse spasm" results. This is an unrecognized high-grade
intestinal obstruction. The hiatus, while adequate to allow the passage of the
esophagus, is too small to contain the fundus and its attachments. Instead of
stretching to accommodate the retracted fundus, it constricts and obstructs
the lumen.
The radiologic appearance described in "diffuse spasm" is no different from
that seen in small bowel obstruction. Rapid, forceful, churning peristalsis
and en masse (tertiary) contractions, that, nevertheless, do not succeed
in clearing the lumen of its contents are typical.
Both the "hiatal squeeze" and the presence of an aperistaltic segment of gut
above the diaphragm prevent esophageal emptying. This tubular segment of fundus
is usually mistaken for esophagus because of the LM tension. Eventually obstruction
results in hypertrophy (diffuse muscular hypertrophy) or in dilatation, decompensation
and elongation of the esophagus.
The diffuse spasm phase may persist for many years. If it does, a blowout of
a weak area in the distal esophagus is likely. This is the epiphrenic diverticulum
- a buffer for the bolus.
Eventually, the circular muscle decompensates and dilates. The LM elongates.
The typical dilated, sigmoid esophagus of "achalasia" does not differ in any
significant way from decompensated bowel proximal to an obstruction elsewhere
in the gut.
Dilatation per se spreads out the ganglion cells like spots on the
surface of an expanding balloon so that there are fewer seen per microscopic
field. Microscopically, this gives the impression of an aganglionosis. As distention
thins the bowel wall it separates the neurons from their end organs, denervating
many of them. The stagnant contents distending the esophageal wall causes first
the neurons and eventually smooth muscle cells to degenerate.
Invasion of the hiatus by tumor or retroperitoneal fibrosis obstructs it causing
"secondary" achalasia. Extirpation of a tumor or chemotherapeutic lysis reverses
typical radiologic and manometric signs of achalasia. Reducing a HH has the
same effect in "primary" achalasia. None of these successful forms of treatment
are directed at either cure of an aganglionosis or "reordering" a disordered
motor function. None of these treatments could conceivably induce regeneration
of lost neurons.
EMDs are not esophageal motor disorders. The manometric and radiographic "abnormalities"
are normal motor responses to obstruction.
The implications for treatment are obvious. The strangulation should be reduced
as soon as the picture of "diffuse spasm" presents. The pulldown operation in
its pure form as employed by Boerema (no fundoplication) is a rational way to
treat the condition. Even if supplemented by enlargment of the hiatus
to prevent strangulation, this should be simpler and safer than the more drastic
means now employed.
"Forceful dilatation" with a bag placed at the hiatal level is effective because
it stretches the hiatus, not the sphincter, but can result in rupture and mediastinitis.
Many other operations with various rationales are effective because exposing
the area of interest reduces the strangulation. If normal peristalsis in not
restored, the open operation should be considered. Laparoscopic surgery could
well prove the best and safest treatment when a procedure is devised.
The Allison procedure - at least as Allison described it(139)
- reduces the size of the hiatus with "non-strangulating sutures." It shares
with fundoplication the potential for insuring obstruction should the original
mechanics cause a recurrence. It is more rational to reduce the strangulated
HH and find means to prevent strangulation if it recurs.
A formidable body of medical research has grown to encrust the aganglionosis
theory of achalasia. It is now being revised to fit the theory that DES and
achalasia are a spectrum of primary muscle disorders. While not without merit,
this work is basically ad hoc, its value dissipated by interpretation
in the light of incorrect hypotheses. Diffuse spasm, idiopathic diffuse muscular
hypertrophy, EMDs and achalasia are but manifestations of the same disorder
- undiagnosed intestinal obstruction. I propose "fundic incarceration" as the
name for all four.
119. Allison, P.R., op cit.
References
Last Updated July 30, 2007 by David PJ Stiennon
1. 1. Hurst, A.F., and Rake, G.W., Achalasia of the cardia (So-called cardiospasm). Quart. J. Med. 23:491, 1930.
2. Achalasia became one of those words like serendipity that always entailed a paragraph or so explaining its derivation.
3. Perhaps stimulated by this problem, researchers are actually measuring action potentials from individual neurons of the myenteric plexus.
4. As reported by Kramer and Ingelfinger, Cannon's Law holds that "...destruction of a unit in a series of efferent neurons is followed by an increased irritability to chemical agents in the isolated structure or structures, the effect being maximal in the part directly denervated."
5. 2. Fisher, R.S., Is Chagasic megaesophagus a model for classical achalasia? Gastroenterology 81: 401-2, 1981.
6. 3. Casella, R.R., Brown, A.L., Jr., Sayre, G. and Ellis, F.H. Jr., Achalasia of the esophagus: pathologic and etiologic considerations. Ann. Surg. 160:474-486, 1964.
7. 4. Misiewicz, J.J., Waller, Schiela L., Anthony, P. and Gummer, J.W., Achalasia of the cardia: pharmacology and histopathology of isolated cardiac sphincteric muscle from patients with and without achalasia. Quart. J. Med. 38:17-30, 1969.
8. 5. Wong, R.K.H., and Johnson, L.F., Achalasia. In: Esophageal function in health and disease. Eds Castell, Donald O. and Johnson, Lawrence F., Elsevier Biomedica, New York, 1983.
9. 6. Postlethwait, R.W., Surgery of the Esophagus, Appleton-Century-Crofts, Norwalk, Connecticut, 1986.
10. 7. Castell, Donald O., The Esophagus, Little Brown and Company, Boston, 1992.
11. 8. Vantrappen, G. and Hellemans, J. In: Diseases of the esophagus. Eds. Cohen, Sidney and Soloway, Roger D., Churchill Livingstone, New York, 1982.
12. 9. Kramer, Philip, Harris, Lauran D. and Donaldson, Robert M., Jr., Transition from symptomatic diffuse spasm to cardiospasm. Gut 8:115-119, 1967.
13. 10. Schroeder, S., Acord, J.L. and Rodgers, J.V., "Achalasia" preceded by non cardiospastic diffuse esophageal spasm. Gastroenterology 44 849, 1963.
14. 11. Barrett, N.R., Achalasia of the cardia: reflections upon a clinical study of over 100 cases. Brit. Med. J., 1:1135-40, 1964.
15. 12. Vantrappen, G., Janssens, J., Hellemans, J. and Coremans, G., Achalasia, diffuse muscle spasm and related motility disorders. Gastroenterology 76:450-457, 1979.
16. In their binary classification, P and p stand for peristalsis present or absent, R and r for sphincter relaxation present or absent and V or v for "vigorous" or not. Although this gives only 8 possibilities, in actuality, there were numerous intermediate disorders. These groups (with transitional forms between them) formed an almost continuous spectrum from diffuse spasm on the active end to achalasia on the passive. Moreover, there was considerable migration from one classification to another especially after forceful dilatation of what they believed to be the sphincter.
17. 13. Ouyang, Ann and Cohen, Sidney, Motor disorders of the esophagus. In: Bockus Gastroenterology, Fourth Edition, Vol. 2, Ed. Berk, J. Edward, W.B. Saunders Company, Philadelphia, 1985.
18. 14. Vantrappen, G and Hellemans, J., Esophageal motor disorders. In: Diseases of the esophagus. Eds. Cohen, Sidney and Soloway, Roger D., Churchill Livingstone, New York, 1982.
19. 15.Ott, D.J., Achalasia, In: Gastrointestinal Radiology, Eds, Gore, Richard M., Levine, Mark S. & Laufer, Igor, Eds., W.B. Saunders, Philadelphia, 1994.
20. 16. Kuhn, The Structure of Scientific Revolutions. University of Chicago Press, Chicago, 1970.
21. 17. Trounce, J.R., Deuchar, D.C., Kauntze, R., and Thomas, G.A., Studies in achalasia of the cardia. Q.J. Med. 26:433-43, 1957.
22. 18. Christensen, James and Robison, Bryce A., Anatomy of the myenteric plexus of the opossum esophagus. Gastroenterology 83: 1033-42, 1982.
23. 19. Earlam, Richard, A vascular cause for Hirschprung's disease? Gastroenterology 88: 1274-9, 1985.
24. 20. Hirschprung's Disease, aganglionic or hypoganglionic megacolon. Am. J. Pathol. 79:189-92, 1975.
25. 21. Raffensperger, John G., Hirschprung's disease. In: Bockus Gastroenterology, Fourth Edition, Vol. 2, Ed. Berk, J. Edward, W.B. Saunders Company, Philadelphia, 1985.
26. 22. Padovan, W., Godoy, R.A., Dantos, R.O. et al., Lower esophageal sphincter response to pentagastrin in chalasic patients with megaesophagus and megacolon. Gut 21: 85-90, 1980.
27. For LESP read hiatal squeeze pressure. The illustrations will show how LMC opens the hiatus as well as the sphincter.
28. 23. Cohen, S. and Lipschutz, W., Lower esophageal sphincter dysfunction in achalasia, Gastroenterology 61:814-20, 1971.
29. 24. Holloway, Richard H., Dodds, Wylie J., Helm, James F., et al. Integrity of cholinergic innervation to the lower esophageal sphincter in achalasia. Gastroenterology 90:924-9, 1986.
30. 25. Orlando, Roy C. and Bozymski, Eugene M., The effects of pentagastrin in achalasia and diffuse esophageal spasm. Gastroenterology 77: 472-7, 1979.
31. 26. Betarello, A. and Pinotti, H.W., Oesophageal involvement in Chagas' disease. Clin. Gastroent.B 5:103-17, 1976.
32. 27. Misiewicz, J.J. et al., op cit..
33. 28. Christensen and Robison, op cit..
34. 29. Paden, P.A., Corkscrew esophagus. U.S. Armed Forces Med. J. 5:1371-74, 1954.
35. 30. Roth, H. and Flescher, B., Diffuse esophageal spasm. Arch. Int. Med. 61:914-23, 1964.
36. 31. Henderson, R.D., Esophageal manometry in clinical investigation. Praeger Publishers, New York, 1983.
37. 32. Mellow, Mark H., Return of esophageal peristalsis in idiopathic achalasia. Gastroenterology 70: 1148-51, 1976.
38. 33. Bennett, John R., Donner, Martin W. and Hendrix, Thomas R., Diffuse esophageal spasm: return to normality. Johns Hopkins Med. J. 126:217-24, 1970.
39. 34. Earlam, Richard, Clinical tests of oesophageal function, Grune & Stratton, New York, 1975.
40. The argument is not entirely valid as neurological disorders affecting widely separated nuclei do exist.
41. 35. Diamant, N. E., Esophageal achalasia (megaesophagus), In: Spontaneous animal models of human disease. Eds, Andres, Edwin J., Ward, Billy C. and Altman, Norman H., Academic Press, New York, 1979.
42. 36. Binder, H.J., Clemett, A.R., Thayer, W.R. and Spiro, H.M., Rarity of hiatus hernia in achalasia. New Eng. J. of Med. 272:680-82, 1965.
43. 37. Allison, P.R., Reflux esophagitis, sliding hiatal hernia and the anatomy of repair. SG&O 92:419-31, 1951.
44. 38. Harrington, S., Discussion of paper of Riegler, Leo G. and Eneboe, John B. 1932
45. 39. Johnson, H. Daintree, Active and passive opening of the cardia in relation to the pathogenesis of hiatus hernia. Gut 7:392-401, 1966.
46. 40. Butler, H., The veins of the esophagus. Thorax 6:276-96, 1951.
47. An unfounded admonition that giving barium to a patient suspected of small bowel obstruction may convert a partial to a complete obstruction limits the opportunities for radiologists to observe barium filled, obstructed bowel.
48. In this operation, the stomach is pulled out of the hiatus and the lesser curvature fixed to the left rectus sheath.
49. This was 1968.
50. 41. Beardsley, J.M. and Thompson, W.R., Acutely obstructed hiatal hernia. Ann. Surg. 159:49, 1964.
51. 42. Bosher, L.H., Fishman, L. and Old, L., Strangulated diaphragmatic hernia with gangrene and perforation of the stomach. Dis. Chest 37:504, 1960.
52. 43. Henderson, Robert D., op cit..
53. 44. Boerema, M.D., Gastropexia anterior geniculata for sliding hiatus hernia and for cardiospasm, J. Internat. Col. of Surg. 29:533-47, 1957.
54. The article is so refreshingly simple, sensible and logical it should be read in the original.
55. 45. Habein, H.C., Moersch, H.J. and Kirklin, J.W., Diverticula of the lower part of the esophagus; a clinical study of one-hundred-forty-nine non-surgical cases. Arch. Intern. Med. 97:768-77, 1956.
56. 46. Harrington, S.W., Surgical treatment of pulsion diverticula of the thoracic esophagus. Ann. Surg. 129:606, 1945.
57. 47. Knuff, T.E. and Castell, D.O., Esophageal diverticula. In: Esophageal function in health and disease. Eds Castell, Donald O. and Johnson, Lawrence F., Elsevier Biomedica, New York, 1983.
58. 48. Effler, D.B., Barr, D.W. and Groves, L.K., Epiphrenic diverticulum of the esophagus, surgical treatment. A.M.A. Arch. Surg. 79:459-67, 1959.
59. 49. Goodman, H.I. and Parnes, I.H., Epiphrenic diverticulum of the esophagus, J. Thoracic. Surg. 23:145-49, 1952.
60. 50. Allen, Thomas H. and Clagett, O. Theron, Changing concepts in the surgical treatment of pulsion diverticula of the lower esophagus. J. Thoracic & Cardiovas. Surg. 50:455-62, 1965.
61. 51. Henderson, Robert D., Esophageal manometry in clinical investigation. Praeger Publishers, New York, 1983.
62. 52. Putney, F. J. and Clerf, Louis H., Epiphrenic esophageal diverticulum. Ann. of Oto. 62:802-17, 1953.
63. It is pretty good evidence against a subphrenic esophageal segment as well. If there were such a segment, these diverticula would be subphrenic.
64. They also occur occasionally in normal patients.
65. 53. Mandelstam, Paul and Lieber, Arthur. Cineradiographic evaluation of the esophagus in normal adults. Gastroenterology 58:32-39, 1970.
66. 54. Nagler, R. and Spiro, H.M., Serial esophageal motility studies in asymptomatic young subjects. Gastroenterology 41:371-9, 1961.
67. 55. Elevator esophagus: fluoroscopic clue to possible organic disease. Radiology 75:914-8, 1960.
68. 56. Stiennon, O. Arthur, The "Captive bolus" test and the pinchcock at the diaphragm: an esophageal pump and some non-diseases of the esophagus. AJR, 99:223-2232, 1967.
69. 57. Mittal, K.K., Bednarz, F. and McCallum, R.W., The acute effects of partial lower esophageal sphincter occlusion on the esophageal motor functions. (Abstract) Gastroenterology 91:1061, 1986.
70. 58. Little, Alex F.G., Fillipo, S. Correnti, et al., Effect of incomplete obstruction on feline esophageal function with a clinical correlation. Surgery 100:430-6, 1986.
71. 59. Kaye, M.D., Anomalies of peristalsis in idiopathic diffuse oesophageal spasm. Gut 22:217-22, 1981.
72. In the small bowel, this type of peristalsis is the cause of the "increased bowel sounds" - a classic physical sign of small bowel obstruction. It would be surprising if the diagnosis of DES could not be made by auscultation of the chest.
73. 60. Bennett, John F. et al., op cit..
74. 61. Kramer, Philip and Ingelfinger, F.J., Esophageal sensitivity to Mecholyl in cardiospasm. Gastroenterology 19:242-51, 1951.
75. 62. Kramer, Philip, Fleshler, Bertram, McNally, Edmund and Harris, Lauran D., Oesophageal sensitivity to Mecholyl in diffuse spasm. Gut 8:120-27, 1967.
76. 63. Goldin, N.R, Burns, T.W., Harrington, J., Treatment of nonspecific esophageal motor disorders. Gastroenterology 82:1069A, 1982.
77. If DES and achalasia are early and late stages of the same disease, the late appearance of the positive Mecholyl test (if we assume that the test does mean denervation) would only tend to show that denervation is a result, not a cause of the disease. A hyperactive response to Mecholyl is not evidence of aganglionosis unless it can be demonstrated that the esophagus is aganglionic in DES. It is not.
78. 64. Vantrappen, op cit..
79. 65. Lamet, M., Fleshler, B. and Achkar, E., Return of peristalsis in treated achalasia: true or technical. (Abstract) Gastroenterology 86:1151, 1984.
80. 66. Bennett, John R., Donner, Martin W. and Hendrix, Thomas R., Diffuse esophageal spasm: return to normality. John Hopkins Med. J. 126:217-24, 1970.
81. 67. Beck, Ivan T., Diagnostic difficulties at the esophagogastric junction. In: The esophagogastric junction. Proceedings of a symposium held in Las Croabas, Puerto Rico, Eds. Katz, David and Hoffman, Fredrick. Excerpta Medica, Princeton, 1971.
82. 68. Morrisey, John F., (Editorial) You see what you look for. Gastroenterology 91:481-2, 1986.
83. 68. Van der Veer, Lindsley D., Krammer, Kenneth, et al., The esophagogastric polyp-fold complex. Am. J. Gastroenter. 79:918-20, 1984.
84. 70. Edwards, D.A.W., Techniques for the radiological examination of the esophagus. In: Disorders of the esophagus: advances and controversies. Eds. Watson, A. and Celestin, L.R., Pitman Publishing, Ltd., London, 1984.
85. 71. Skinner, David B., Klementschitsch, Peter, Little, Alex G., DeMeester, Tom, and Belsey, Ronald R.H., Assessment of failed antireflex repairs. In: Esophageal Disorders, Eds. DeMeester, Tom R. and Skinner, David B., Raven Press, New York, 1985.
86. 72. Kumar, R. A case of esophageal stricture due to a cartilaginous ring. Brit. J. Surg. 49:543-4, 1962.
87. 73. Seaman, W.B., Wells, J. and Flood, C., Diagnostic problems of esophageal cancer: relationship to achalasia and hiatus hernia. Am J. Roentgenol. Rad. Therapy & Nuclear Med. 90:778-91, 1963.
88. As the Mecholyl test has been positive in patients whose achalasia cleared after relief of a hiatal obstruction, it cannot exemplify Cannon's Law. It does seem to be diagnostic of esophageal obstruction. This leads to a "catch-22" situation: the test is positive for a condition in which it should be contraindicated.
89. 74. Herrara, A.F., Colon, J., Valdes-Dapena and Roth, J.L.A. Achalasia or carcinoma? The significance of the Mecholyl test. Am. J. Dig. Dis. 15:1073-81, 1970
90. 75. Mellow, Mark H., op cit..
91. 76. Ferguson, Thomas B. and Burford, Thomas H., An evaluation of the modified Heller operation in the treatment of achalasia of the esophagus. Ann. Surg. 152:1-9, 1960.
92. 77. Tucker, Harold J., Snape, William J., Jr. and Cohen, Sidney, Achalasia secondary to carcinoma: manometric and clinical features. Ann. Int. Med. 89:315-18, 1978.
93. 78. Sandler, Robert S., Bozymski, Eugene M. and Orlando, Roy C., Failure of clinical criteria to distinguish between primary achalasia and achalasia due to tumor, Dig. Dis. Sci. 27:209-13, 1982.
94. 79. Schultz, K.S., Goresky, C.A., Jabbari, M. and Lough, J., Esophageal achalasia associated with gastric carcinoma: lack of evidence for widespread plexus destruction.
95. 80. Tucker, Harold J. et al., op cit..
96. 81. Davis, John A., Kantrowitz, Paul A., Chandler, Harold L. and Schatzki, Stefan C., Reversible achalasia due to reticulum cell sarcoma, New Eng. J. of Med. 293:130-2, 1979.
97. 82. Kline, Michael M., Successful treatment of vigorous achalasia associated with gastric lymphoma. Dig. Dis. Sci. 25:311-13, 1971.
98. 83. Nelson, Russell M., Jenson, Conrad B., Horsley, Brent L. and Ershler, Irving, Idiopathic retroperitoneal fibrosis producing distal esophageal obstruction. J. Thoracic & Cardiovas. Surg. 55:216-24, 1968.
99. 84. Dodds, Wiley J., Stewart, Edward T., Kishk, Soad M., et al. Radiologic amyl nitrite test for distinguishing pseudoachalasia from idiopathic achalasia. AJR 146:21-3, 1986.
100. 85.Menin, Richard and Fisher, Robert S., Return of esophageal peristalsis in achalasia secondary to gastric cancer. Dig. Dis. Sci. 56:1038-44, 1981.
101. 86. Miller, R.H., Amyloid disease - an unusual cause of megalo-esophagus. South African Med. J. 45:1202-3, 1969.
102. 87. Lefkowitz, J.R., Brugge, W.R., Schuffler, M.D. and Brand, D.L., (Abstract) Amyloidosis mimics achalasia's effect on the lower esophageal sphincter. Gastroenterology 91:1059, 1986.
103. 88. Pierandozzi,J.S., Ritter, J.H., Transient achalasia:a complication of vagotomy. Am. J. Surg. 111:356-58, 1966.
104. 89. Leonardi, Howard K. and Ellis, F. Henry, Jr, Complications of Nissen Fundoplication. Surg. Clin. N. Amer. 63:1155-65, 1983.
105. 90. Kenney, R., Levine, M., Mintz, M. and Cohen, S., Diagnostic utility of computed tomography in tumor associated (secondary) achalasia. (Abstract) Gastroenterology 90:1801, 1986.
106. Clinical physiologists tend to believe the LES is 3-5 cm in length. This could be due to the low resolving power of the manometer, to respiratory motion or measuring hiatal squeeze rather than the sphincter.
107. 91. Gershon, Michael D. and Erde, Steven M., The nervous system of the gut. Gastroenterology 80:1571-94, 1981.
108. 92. You, Chul H., Lee, K.Y., Chey, William Y. and Menguy, Rene, Electrogastrographic study of patients with unexplained nausea, bloating and vomiting. Gastroenterology 79:311-4, 1980.
109. 93. Katz, P.O., Richter, J.E., Dalton, C.B., et al., "Incomplete" lower esophageal sphincter relaxation in achalasia: an absolute requirement? (Abstract) Gastroenterology 88:1439, 1985.
110. 94. Mittal, Ravinder K., Lang, Robert C. and McCallum, Richard W. Gastroenterology 92:130-5, 1987
111. 95. Feczko, Peter J. and Halpert, Robert D., Achalasia secondary to non-gastrointestinal malignancies. Gastrointest. Rad. 10:273-6, 1985.
112. The persistence of some of these pseudotumors after reduction suggests that in some cases adhesions may occur during an incarceration.
113. 96. Henderson, Robert D. and Godden, John O., Motor disorders of the Esophagus Williams & Wilkins, Baltimore, 1976.
114. 97. Johnstone, A.D., Diffuse spasm and diffuse muscular hypertrophy of the lower esophagus. Brit. J. Radiol. 33:723-35, 1960.
115. 98. Blank, Eugene and Michael, Thomas D., Muscular hypertrophy of the esophagus: report of a case with involvement of the entire esophagus. Pediatrics 32:595-8, 1963.
116. 99. Sloper, J.C., Idiopathic diffuse muscular hypertrophy of the lower esophagus. Thorax 9:136-46, 1954.
117. 100. Spencer, Rowena and Hudson, Thomas L., Idiopathic muscular hypertrophy of the gastrointestinal tract in a child. Pediatr. Surg. 50:678-86, 1961.
118. 101. Zeller, Robert, McClelland, Robert, Meyers, Beverly, et al., Idiopathic muscular hypertrophy of the esophagus, Gastrointest. Radiol. 4:121-5, 1979.
119. This effect has probably caused the epidemic of cases of Barrett esophagus now appearing in the literature. If a surgeon can mistake a tubular fundus for esophagus, it is probable that an endoscopist will have the same problem. To prove such a diagnosis - which Barrett, himself, originally doubted - it would be necessary to have angiographic evidence that the blood supply to that region of the "esophagus lined with gastric mucosa" originated above the diaphragm.
120. It is significant that in these obstructions the hypertrophy increases progressively in the more distal portions of the esophagus. This is because there is increasingly more work involved as the pressure ahead of the p-wave builds.
121. 102. Qualman, Stephan J., Haupt, Helen M., Yang, Peter, et al., Esophageal Lewy bodies associated with ganglion cell loss in achalasia. Gastroenterology 87:848-56, 1984.
122. 103. Cohen, Bernard R., Cardiospasm in achalasia. Gastroenterology 48:864-9, 1965.
123. 104. Cohen, Bernard R., Chapter 1 In: The esophagogastric junction. Proceedings of a symposium held in Las Croabas, Puerto Rico, Eds. Katz, David and Hoffman, Fredrick. Excerpta Medica, Princeton, 1971.
124. 105. Cohen, Bernard R., "Cardiospasm" in achalasia:demonstration of an abnormally elevated esophagogastric sphincter pressure with partial relaxation on swallowing. Gastroenterological research forum: abstracts 48: 864, 1965.
125. 106. Weihrauch, Thomas R., Esophageal Manometry. Urban & Schwartzenberg, Baltimore, 1981.
126. 107. Katz, Philip O., Richter, Joel E., Cowan, Robert and Castell, Donald O., Apparent complete lower esophageal sphincter relaxation in achalasia. Gastroenterology 90:978-83, 1986.
127. 108. Vantrappen, G and Hellmans, J., Treatment of achalasia and related motor disorders. Gastroenterology 79:144-54, 1980.
128. 109. Vantrappen, G. and Hellmans, J., 1980, op. cit..
129. 110. Surgery for esophageal motor disorders, Demeester, Tom R. and Stein, Herbert J., In: The Esophagus, Ed. Castell, Donald O., Little Brown and Company, Boston, 1992.
130. 111. Black, J., Vorbach, A.N. and Collis, J., Results of Heller operation for achalasia of the esophagus: the importance of hiatal hernia. Brit. J. Surg. 63:949-53, 1976.
131. 112. Okike, Nsidinanya, Payne, W. Spencer, Neufeld, David M., Bernatz, Philip E., Pairolero, Peter C. and Sanderson, David R., Esophagomyotomy versus forceful dilation for achalasia of the esophagus: results in 899 patients. Ann. Thorac. Surgery 28:119-25, 1979.
132. 113. Euphrat, Edwin J. and Delmonico, J. Earnest, Jr., Coexistent achalasia of the esophagus and hiatal hernia of the cardiac end of the stomach. J. Thoracic Surg. 34:395-8, 1957.
133. 114. Furguson, Thomas B. and Burford, Thomas H., Op. cit..
134. Although it is doubtful that any surgeon would agree with Binder et al. that HH and achalasia do not occur together, and the exact opposite is true, their article made a key contribution to this solution of the puzzle because it proved that even on the most careful study passed by referees, HH's went undetected in the presence of achalasia.
135. 115. Ellis, F. Henry, Jr., Gibb, S. Peter and Crozier, Robert E., Esophagomyotomy for achalasia of the esophagus. Ann. Surg. 193:157-61, 1980.
136. 116. Wong, R.H.K and Johnson, L.F., op cit..
137. 117. Trounce, J., et al., op cit..
138. 118. Boerema, op. cit..
139. 119. Allison, P.R., op cit.